CDMO Biologics Manufacturers in karnataka
Our research team comprises of experts in molecular biology, biochemistry, process characterization, scale-up activities, and fill-finish developments.
Our best-in-class drug substance/drug product development and manufacturing space, with R&D, analytical development, manufacturing, and quality control labs making us the right size to deliver on all of your project needs. Our expert team has vast experience to help you take your product from concept to commercialization. The Quality and Regulatory teams provide added support during the critical manufacturing processes.
Small Volume manufacturing
50Ltrs and 200Ltrs bioreactors (with and without perfusion)
Large Volume manufacturing
2 x 2000Ltrs Bioreactors and 4 x 1000Ltrs Bioreactor (total 8000 Ltrs)
Planned expansion
16000 Ltrs capability
Small Volume manufacturing
20 units/m (PFS and vial)
Large Volume manufacturing
40 units/m, Isolator (PFS and vial)
High Volume manufacturing
300 units/m vial filling (Size 2R to 100R)
PFS Plunger rod assembly& labelling machine
80 units / min.
PFS/vial blister packing machine
80 units / min.
Vial Stricker labelling machine
130 units / min.
Biosimilar development Manufacturers in karnataka
Biosimilars are attractive for their affordable costs and access enhancement to a wider patient population. The world has witnessed a growing need for biosimilars treatment and the market is expected to grow enormously in the next decade. ShilpaBio welcomes partnership opportunities to maximize the biosimilars reach to a diverse patient population across globe.
As the name suggests, Biosimilars signify the similarity to original biologic medicines providing the same quality, efficacy and safety evaluated by regulatory agencies
A patient treated with a reference product can change to its approved biosimilar, without loss of efficacy or an increase in safety risk along with an added benefit of costs savings
Biosimilars offer promises to the healthcare professionals with,
1) More treatment options
2) More access to medications that can save lives
3) Possibility that market-competition will drive down the costs of healthcareBiosimilars approval enables interchangeability of medicines in the hope of increased access to important and life-saving medications to the global patient population
albumin characterization in India
Albumin is the most abundant protein in human blood, accounting for 50 to 60% of all the protein in the blood. It’s made by the liver, circulates throughout the body in blood, and a very small amount may pass through healthy kidneys into urine.
Albumin’s critical functions in maintaining osmotic pressure, binding of blood substances, including its anti-oxidant potential is well documented.
Inspired by the animal-free origin and consistent nature of recombinant products, Shilpa Biologics team has innovated a recombinant version of Albumin that is manufactured from a complete synthetic process having many advantages. NavAlbumin is our flagship product that has the potential to expand to diverse applications. The therapeutic community has just recently started to fully comprehend the potential that it provides.
ShilpaBio is currently scaling up the volumes to 150KL, and the new facility will add high capability to position us as the world’s leading rAlbumin producer by volume.

CDMO Biologics Manufacturers in Arunachal Pradesh
Our research team comprises of experts in molecular biology, biochemistry, process characterization, scale-up activities, and fill-finish developments.
Our best-in-class drug substance/drug product development and manufacturing space, with R&D, analytical development, manufacturing, and quality control labs making us the right size to deliver on all of your project needs. Our expert team has vast experience to help you take your product from concept to commercialization. The Quality and Regulatory teams provide added support during the critical manufacturing processes.
Small Volume manufacturing
50Ltrs and 200Ltrs bioreactors (with and without perfusion)
Large Volume manufacturing
2 x 2000Ltrs Bioreactors and 4 x 1000Ltrs Bioreactor (total 8000 Ltrs)
Planned expansion
16000 Ltrs capability
Small Volume manufacturing
20 units/m (PFS and vial)
Large Volume manufacturing
40 units/m, Isolator (PFS and vial)
High Volume manufacturing
300 units/m vial filling (Size 2R to 100R)
PFS Plunger rod assembly& labelling machine
80 units / min.
PFS/vial blister packing machine
80 units / min.
Vial Stricker labelling machine
130 units / min.
Bioprocess R&D in India
Small Volume manufacturing
High operation costs of cell culture media
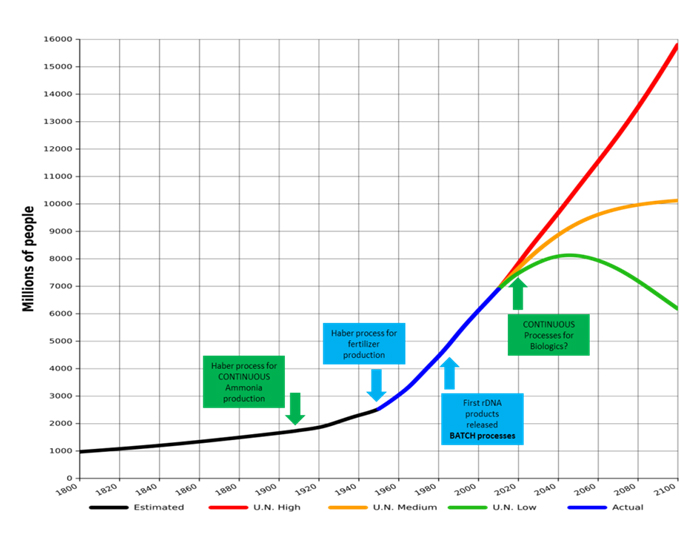
High density continuous bioprocess platform for production of biopharmaceuticals from small footprint facility - ContiMAb™ , ContiFuse™ & ContiVir™
Development of low-cost cell culture media and media kit to enable continuous processing
Continuous bioprocess platform ContiMAb & ContiFuse platforms to enable penetration of biologics from 5% to 30% of the patient population. Our scientists leverage the platforms for biosimilars portfolio development.
We welcome partners who share the mutual goal of making high quality biopharmaceuticals affordable to patients globally.
Please write to bd_bio@shilpabio.com with your queries.
 Shilpa Biologicals Pvt Ltd
Shilpa Biologicals Pvt LtdPlot 532/A, Belur Industrial Area,
Dharwad-580011
Karnataka INDIA
Call us: +91- 836 248 5555
Email: info@shilpabio.com
CDMO Biologics Manufacturers in Assam
Our research team comprises of experts in molecular biology, biochemistry, process characterization, scale-up activities, and fill-finish developments.
Our best-in-class drug substance/drug product development and manufacturing space, with R&D, analytical development, manufacturing, and quality control labs making us the right size to deliver on all of your project needs. Our expert team has vast experience to help you take your product from concept to commercialization. The Quality and Regulatory teams provide added support during the critical manufacturing processes.
Small Volume manufacturing
50Ltrs and 200Ltrs bioreactors (with and without perfusion)
Large Volume manufacturing
2 x 2000Ltrs Bioreactors and 4 x 1000Ltrs Bioreactor (total 8000 Ltrs)
Planned expansion
16000 Ltrs capability
Small Volume manufacturing
20 units/m (PFS and vial)
Large Volume manufacturing
40 units/m, Isolator (PFS and vial)
High Volume manufacturing
300 units/m vial filling (Size 2R to 100R)
PFS Plunger rod assembly& labelling machine
80 units / min.
PFS/vial blister packing machine
80 units / min.
Vial Stricker labelling machine
130 units / min.

Plot 532/A, Belur Industrial Area,
Dharwad-580011
Karnataka INDIA.
Biosimilars Manufacturers in karnataka
Biosimilars are attractive for their affordable costs and access enhancement to a wider patient population. The world has witnessed a growing need for biosimilars treatment and the market is expected to grow enormously in the next decade. ShilpaBio welcomes partnership opportunities to maximize the biosimilars reach to a diverse patient population across globe.
As the name suggests, Biosimilars signify the similarity to original biologic medicines providing the same quality, efficacy and safety evaluated by regulatory agencies
A patient treated with a reference product can change to its approved biosimilar, without loss of efficacy or an increase in safety risk along with an added benefit of costs savings
Biosimilars offer promises to the healthcare professionals with,
1) More treatment options
2) More access to medications that can save lives
3) Possibility that market-competition will drive down the costs of healthcareBiosimilars approval enables interchangeability of medicines in the hope of increased access to important and life-saving medications to the global patient population

Plot 532/A, Belur Industrial Area,
Dharwad-580011
Karnataka INDIA.
Albumin is the most abundant protein in human blood, accounting for 50 to 60% of all the protein in the blood. It’s made by the liver, circulates throughout the body in blood, and a very small amount may pass through healthy kidneys into urine.
Albumin’s critical functions in maintaining osmotic pressure, binding of blood substances, including its anti-oxidant potential is well documented.
Inspired by the animal-free origin and consistent nature of recombinant products, Shilpa Biologics team has innovated a recombinant version of Albumin that is manufactured from a complete synthetic process having many advantages. NavAlbumin is our flagship product that has the potential to expand to diverse applications. The therapeutic community has just recently started to fully comprehend the potential that it provides.
ShilpaBio is currently scaling up the volumes to 150KL, and the new facility will add high capability to position us as the world’s leading rAlbumin producer by volume.

Plot 532/A, Belur Industrial Area,
Dharwad-580011
Karnataka INDIA.
Albumin-is-the-most...." >
CDMO Biologics Manufacturers in Karnataka
Our research team comprises of experts in molecular biology, biochemistry, process characterization, scale-up activities, and fill-finish developments.
Our best-in-class drug substance/drug product development and manufacturing space, with R&D, analytical development, manufacturing, and quality control labs making us the right size to deliver on all of your project needs. Our expert team has vast experience to help you take your product from concept to commercialization. The Quality and Regulatory teams provide added support during the critical manufacturing processes.
Small Volume manufacturing
50Ltrs and 200Ltrs bioreactors (with and without perfusion)
Large Volume manufacturing
2 x 2000Ltrs Bioreactors and 4 x 1000Ltrs Bioreactor (total 8000 Ltrs)
Planned expansion
16000 Ltrs capability
Small Volume manufacturing
20 units/m (PFS and vial)
Large Volume manufacturing
40 units/m, Isolator (PFS and vial)
High Volume manufacturing
300 units/m vial filling (Size 2R to 100R)
PFS Plunger rod assembly& labelling machine
80 units / min.
PFS/vial blister packing machine
80 units / min.
Vial Stricker labelling machine
130 units / min.
novel biologics manufacturers in karnataka
Recombinant NavAlbumin could be explored for creative formulation strategies to customize based on precise needs. Our scientific experts would work with you closely to develop therapeutic solutions to scale up into a commercial product.
NavAlbumin can reduce costs down the line by increasing safety and reducing the patient inconvenience at the hospital level. If NavAlbumin is used early in the development cycle, it can solve stability issues that would previously have delayed product launch.
' NavAlbumin is a cost effective, single solution to many stability issues, and is particularly effective when used in very early development – products can be stabilized quickly in a common formulation, speeding up the long route to market.

Biosimilars Manufacturers in Andhra Pradesh
Biosimilars are attractive for their affordable costs and access enhancement to a wider patient population. The world has witnessed a growing need for biosimilars treatment and the market is expected to grow enormously in the next decade. ShilpaBio welcomes partnership opportunities to maximize the biosimilars reach to a diverse patient population across globe.
As the name suggests, Biosimilars signify the similarity to original biologic medicines providing the same quality, efficacy and safety evaluated by regulatory agencies
A patient treated with a reference product can change to its approved biosimilar, without loss of efficacy or an increase in safety risk along with an added benefit of costs savings
Biosimilars offer promises to the healthcare professionals with,
1) More treatment options
2) More access to medications that can save lives
3) Possibility that market-competition will drive down the costs of healthcareBiosimilars approval enables interchangeability of medicines in the hope of increased access to important and life-saving medications to the global patient population
CDMO Biologics Manufacturers in Arunachal Pradesh
Our research team comprises of experts in molecular biology, biochemistry, process characterization, scale-up activities, and fill-finish developments.
Our best-in-class drug substance/drug product development and manufacturing space, with R&D, analytical development, manufacturing, and quality control labs making us the right size to deliver on all of your project needs. Our expert team has vast experience to help you take your product from concept to commercialization. The Quality and Regulatory teams provide added support during the critical manufacturing processes.
Small Volume manufacturing
50Ltrs and 200Ltrs bioreactors (with and without perfusion)
Large Volume manufacturing
2 x 2000Ltrs Bioreactors and 4 x 1000Ltrs Bioreactor (total 8000 Ltrs)
Planned expansion
16000 Ltrs capability
Small Volume manufacturing
20 units/m (PFS and vial)
Large Volume manufacturing
40 units/m, Isolator (PFS and vial)
High Volume manufacturing
300 units/m vial filling (Size 2R to 100R)
PFS Plunger rod assembly& labelling machine
80 units / min.
PFS/vial blister packing machine
80 units / min.
Vial Stricker labelling machine
130 units / min.
Bioprocess R&D in karnataka
Small Volume manufacturing
High operation costs of cell culture media

High density continuous bioprocess platform for production of biopharmaceuticals from small footprint facility - ContiMAb™ , ContiFuse™ & ContiVir™
Development of low-cost cell culture media and media kit to enable continuous processing
Continuous bioprocess platform ContiMAb & ContiFuse platforms to enable penetration of biologics from 5% to 30% of the patient population. Our scientists leverage the platforms for biosimilars portfolio development.
We welcome partners who share the mutual goal of making high quality biopharmaceuticals affordable to patients globally.
Please write to bd_bio@shilpabio.com with your queries.
 Shilpa Biologicals Pvt Ltd
Shilpa Biologicals Pvt LtdPlot 532/A, Belur Industrial Area,
Dharwad-580011
Karnataka INDIA
Call us: +91- 836 248 5555
Email: info@shilpabio.com
Biosimilar development Manufacturers in Andhra Pradesh
Biosimilars are attractive for their affordable costs and access enhancement to a wider patient population. The world has witnessed a growing need for biosimilars treatment and the market is expected to grow enormously in the next decade. ShilpaBio welcomes partnership opportunities to maximize the biosimilars reach to a diverse patient population across globe.
As the name suggests, Biosimilars signify the similarity to original biologic medicines providing the same quality, efficacy and safety evaluated by regulatory agencies
A patient treated with a reference product can change to its approved biosimilar, without loss of efficacy or an increase in safety risk along with an added benefit of costs savings
Biosimilars offer promises to the healthcare professionals with,
1) More treatment options
2) More access to medications that can save lives
3) Possibility that market-competition will drive down the costs of healthcareBiosimilars approval enables interchangeability of medicines in the hope of increased access to important and life-saving medications to the global patient population
CDMO Biologics Manufacturers in karnataka
Our research team comprises of experts in molecular biology, biochemistry, process characterization, scale-up activities, and fill-finish developments.
Our best-in-class drug substance/drug product development and manufacturing space, with R&D, analytical development, manufacturing, and quality control labs making us the right size to deliver on all of your project needs. Our expert team has vast experience to help you take your product from concept to commercialization. The Quality and Regulatory teams provide added support during the critical manufacturing processes.
Small Volume manufacturing
50Ltrs and 200Ltrs bioreactors (with and without perfusion)
Large Volume manufacturing
2 x 2000Ltrs Bioreactors and 4 x 1000Ltrs Bioreactor (total 8000 Ltrs)
Planned expansion
16000 Ltrs capability
Small Volume manufacturing
20 units/m (PFS and vial)
Large Volume manufacturing
40 units/m, Isolator (PFS and vial)
High Volume manufacturing
300 units/m vial filling (Size 2R to 100R)
PFS Plunger rod assembly& labelling machine
80 units / min.
PFS/vial blister packing machine
80 units / min.
Vial Stricker labelling machine
130 units / min.
Biosimilar development Manufacturers in karnataka
Biosimilars are attractive for their affordable costs and access enhancement to a wider patient population. The world has witnessed a growing need for biosimilars treatment and the market is expected to grow enormously in the next decade. ShilpaBio welcomes partnership opportunities to maximize the biosimilars reach to a diverse patient population across globe.
As the name suggests, Biosimilars signify the similarity to original biologic medicines providing the same quality, efficacy and safety evaluated by regulatory agencies
A patient treated with a reference product can change to its approved biosimilar, without loss of efficacy or an increase in safety risk along with an added benefit of costs savings
Biosimilars offer promises to the healthcare professionals with,
1) More treatment options
2) More access to medications that can save lives
3) Possibility that market-competition will drive down the costs of healthcareBiosimilars approval enables interchangeability of medicines in the hope of increased access to important and life-saving medications to the global patient population
Albumin is the most abundant protein in human blood, accounting for 50 to 60% of all the protein in the blood. It’s made by the liver, circulates throughout the body in blood, and a very small amount may pass through healthy kidneys into urine.
Albumin’s critical functions in maintaining osmotic pressure, binding of blood substances, including its anti-oxidant potential is well documented.
Inspired by the animal-free origin and consistent nature of recombinant products, Shilpa Biologics team has innovated a recombinant version of Albumin that is manufactured from a complete synthetic process having many advantages. NavAlbumin is our flagship product that has the potential to expand to diverse applications. The therapeutic community has just recently started to fully comprehend the potential that it provides.
ShilpaBio is currently scaling up the volumes to 150KL, and the new facility will add high capability to position us as the world’s leading rAlbumin producer by volume.

Plot 532/A, Belur Industrial Area,
Dharwad-580011
Karnataka INDIA.
Albumin-is-the-most...." >
CDMO Biologics Manufacturers in karnataka
Our research team comprises of experts in molecular biology, biochemistry, process characterization, scale-up activities, and fill-finish developments.
Our best-in-class drug substance/drug product development and manufacturing space, with R&D, analytical development, manufacturing, and quality control labs making us the right size to deliver on all of your project needs. Our expert team has vast experience to help you take your product from concept to commercialization. The Quality and Regulatory teams provide added support during the critical manufacturing processes.
Small Volume manufacturing
50Ltrs and 200Ltrs bioreactors (with and without perfusion)
Large Volume manufacturing
2 x 2000Ltrs Bioreactors and 4 x 1000Ltrs Bioreactor (total 8000 Ltrs)
Planned expansion
16000 Ltrs capability
Small Volume manufacturing
20 units/m (PFS and vial)
Large Volume manufacturing
40 units/m, Isolator (PFS and vial)
High Volume manufacturing
300 units/m vial filling (Size 2R to 100R)
PFS Plunger rod assembly& labelling machine
80 units / min.
PFS/vial blister packing machine
80 units / min.
Vial Stricker labelling machine
130 units / min.
CDMO Biologics Manufacturers in karnataka
Our research team comprises of experts in molecular biology, biochemistry, process characterization, scale-up activities, and fill-finish developments.
Our best-in-class drug substance/drug product development and manufacturing space, with R&D, analytical development, manufacturing, and quality control labs making us the right size to deliver on all of your project needs. Our expert team has vast experience to help you take your product from concept to commercialization. The Quality and Regulatory teams provide added support during the critical manufacturing processes.
Small Volume manufacturing
50Ltrs and 200Ltrs bioreactors (with and without perfusion)
Large Volume manufacturing
2 x 2000Ltrs Bioreactors and 4 x 1000Ltrs Bioreactor (total 8000 Ltrs)
Planned expansion
16000 Ltrs capability
Small Volume manufacturing
20 units/m (PFS and vial)
Large Volume manufacturing
40 units/m, Isolator (PFS and vial)
High Volume manufacturing
300 units/m vial filling (Size 2R to 100R)
PFS Plunger rod assembly& labelling machine
80 units / min.
PFS/vial blister packing machine
80 units / min.
Vial Stricker labelling machine
130 units / min.
Darbepoetin manufacturers in India.
Darbepoetin alfa is a synthetic form of erythropoietin, a glycoprotein hormone that stimulates the production of red blood cells in the bone marrow. It is used to treat anemia, particularly in patients with chronic kidney disease, cancer patients undergoing chemotherapy,
shilpa biologicals company is one the best Darbepoetin manufacturers in India.

Plot 532/A, Belur Industrial Area,
Dharwad-580011
Karnataka INDIA.
CDMO Biologics Manufacturers in Goa
Our research team comprises of experts in molecular biology, biochemistry, process characterization, scale-up activities, and fill-finish developments.
Our best-in-class drug substance/drug product development and manufacturing space, with R&D, analytical development, manufacturing, and quality control labs making us the right size to deliver on all of your project needs. Our expert team has vast experience to help you take your product from concept to commercialization. The Quality and Regulatory teams provide added support during the critical manufacturing processes.
Small Volume manufacturing
50Ltrs and 200Ltrs bioreactors (with and without perfusion)
Large Volume manufacturing
2 x 2000Ltrs Bioreactors and 4 x 1000Ltrs Bioreactor (total 8000 Ltrs)
Planned expansion
16000 Ltrs capability
Small Volume manufacturing
20 units/m (PFS and vial)
Large Volume manufacturing
40 units/m, Isolator (PFS and vial)
High Volume manufacturing
300 units/m vial filling (Size 2R to 100R)
PFS Plunger rod assembly& labelling machine
80 units / min.
PFS/vial blister packing machine
80 units / min.
Vial Stricker labelling machine
130 units / min.
novel biologics manufacturers in Arunachal Pradesh
Recombinant NavAlbumin could be explored for creative formulation strategies to customize based on precise needs. Our scientific experts would work with you closely to develop therapeutic solutions to scale up into a commercial product.
NavAlbumin can reduce costs down the line by increasing safety and reducing the patient inconvenience at the hospital level. If NavAlbumin is used early in the development cycle, it can solve stability issues that would previously have delayed product launch.
' NavAlbumin is a cost effective, single solution to many stability issues, and is particularly effective when used in very early development – products can be stabilized quickly in a common formulation, speeding up the long route to market.
Biologics Manufacturers in Arunachal Pradesh
Welcome to Shilpa Biologicals: Your Trusted Biologics Manufacturers in Arunachal Pradesh
Unlocking the Potential of Curative Therapies at Our State-of-the-Art R&D Facility
Shilpa Biologicals, headquartered in Dharwad, Karnataka, stands as a beacon of innovation in the realm of biologics manufacturing. As a leading player in the industry, we are dedicated to developing cutting-edge curative therapies that address the critical needs of patients worldwide.
Our Commitment to Excellence:
At Shilpa Biologicals, we pride ourselves on a powerhouse team of scientists and professionals who relentlessly pursue scientific breakthroughs. Our R&D manufacturing facility, located at Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India, serves as the epicenter of our commitment to excellence.
Immunology and Haematology Focus:
Our product pipeline is strategically aligned with the immunology and haematology sectors, aiming to make life-changing therapies affordable and accessible to patients globally. With a robust foundation in molecular biology, biochemistry, immunology, and formulations, we are poised to revolutionize the blood-product segment.
Global Competence, Local Presence:
Shilpa Biologicals prides itself on being a global contender for best-in-class biologics manufacturing and services. Our wholly owned projects leverage our deep scientific confidence, allowing us to compete on the global stage while providing cost-effective solutions for patients worldwide.
Key Features of Shilpa Biologicals:
Biosimilars Manufacturers in karnataka
Welcome to Shilpa Biological, a pioneering company based in Karnataka, dedicated to advancing healthcare accessibility through our cutting-edge biosimilars. As a leader in the field, we understand the crucial role affordability plays in ensuring widespread patient access to high-quality treatments.
Our Biosimilars, crafted at our state-of-the-art manufacturing facility located at Plot 532/A, Belur Industrial Area, Dharwad-580011, represent a commitment to excellence and innovation. With an eye on the future, we anticipate a significant surge in the demand for biosimilars, driven by the need for cost-effective solutions that cater to a diverse patient population.
At ShilpaBio, we recognize the importance of strategic partnerships in expanding the reach of our biosimilars across the globe. We invite collaboration opportunities to maximize our impact on a wider demographic and contribute to the global healthcare landscape.
As the biosimilars market is poised for substantial growth in the next decade, Shilpa Biological is poised to be at the forefront of this transformative wave. Join us in our mission to make advanced healthcare solutions accessible to all, ensuring a healthier and more equitable world. Explore partnership possibilities with Shilpa Biological and be part of the journey towards a healthier future.
CDMO Biologics Manufacturers in karnataka
Welcome to Shilpa Biological, your trusted partner in CDMO (Contract Development and Manufacturing Organization) Biologics. Located in the heart of Karnataka, our cutting-edge facility at Plot 532/A, Belur Industrial Area, Dharwad-580011, is dedicated to meeting the growing demand for high-quality and affordable biosimilars.
In an era where biosimilars play a pivotal role in enhancing access to healthcare, ShilpaBio stands out as a beacon of excellence. Our commitment to providing cost-effective solutions aligns seamlessly with the global need for accessible and innovative biologic treatments.
As the world witnesses an escalating demand for biosimilars, the market is poised for significant expansion in the next decade. ShilpaBio, as a key player in this transformative landscape, welcomes collaboration opportunities to amplify the reach of our CDMO Biologics. Our goal is to cater to a diverse patient population worldwide, ensuring that advanced healthcare is within everyone's reach.
Partner with Shilpa Biological to navigate the evolving biosimilars market successfully. By joining forces with us, you contribute to the realization of a healthier and more inclusive future. Explore the possibilities of collaboration with Shilpa Biological, your trusted partner in CDMO Biologics Manufacturing in Karnataka. Together, let's pave the way for accessible and cost-effective healthcare solutions on a global scale.
CMO Biologics Manufacturers in Bihar
Looking for top-tier CMO biologics manufacturers in Bihar? Look no further than Shilpa Biologicals, a leading name in the field of biologics manufacturing and services. Located in Plot 532/A, Belur Industrial Area, Dharwad, Karnataka, India, Shilpa Biologicals boasts a state-of-the-art R&D manufacturing facility dedicated to developing curative therapies.
About Shilpa Biologicals:
At Shilpa Biologicals, we pride ourselves on our cutting-edge team of scientists and committed professionals who work tirelessly on the manufacturing floor. Our primary focus lies in the immunology and haematology sectors, with a mission to make life-saving treatments affordable and accessible to patients worldwide.
Our Commitment to Innovation:
With a firm belief in our scientific prowess, Shilpa Biologicals is dedicated to pushing the boundaries of medical science. We are driven by a deep confidence in our abilities to contribute to high-level scientific breakthroughs, both locally and globally.
Our Expertise:
Drawing upon our expertise in molecular biology, biochemistry, immunology, and formulations, we have developed a robust pipeline of wholly owned projects. These projects have the potential to revolutionize the blood-product segment and compete on a global scale, offering best-in-class services.
Why Choose Shilpa Biologicals:
For inquiries and collaborations, reach out to us at:
Partner with Shilpa Biologicals today and join us in our mission to revolutionize biologics manufacturing for the betterment of patients everywhere.
Stability Testing Of Pharmaceutical Products in Arunachal Pradesh
Are you seeking reliable stability testing services for pharmaceutical products in Arunachal Pradesh? Look no further than Shilpa Biologicals, a trusted name renowned for its commitment to excellence. With our cutting-edge R&D manufacturing facility located at Plot 532/A, Belur Industrial Area, Dharwad, Karnataka, India, we bring forth a wealth of expertise and innovation to the forefront of biologics manufacturing.
Why Choose Shilpa Biologicals for Stability Testing?
At Shilpa Biologicals, we understand the critical importance of stability testing in upholding the integrity and efficacy of pharmaceutical products. Our dedicated team of scientists and professionals works tirelessly to ensure that every medication meets the highest standards of quality and safety.
Tailored Solutions for Arunachal Pradesh:
Recognizing the unique environmental challenges of Arunachal Pradesh, we customize our stability testing protocols to suit the region's specific needs. By leveraging our expertise in molecular biology, biochemistry, and immunology, we deliver comprehensive testing services tailored to the climatic conditions of Arunachal Pradesh, thereby ensuring the reliability and potency of pharmaceutical products.
Unrivaled Expertise and Innovation:
With a focus on developing curative therapies in the immunology and haematology sectors, Shilpa Biologicals stands at the forefront of scientific innovation. Our team of talented professionals is equipped with the knowledge and creativity to address complex scientific challenges and drive groundbreaking advancements in healthcare.
Committed to Patient Welfare:
At Shilpa Biologicals, our ultimate goal is to make life-saving medications accessible and affordable to patients worldwide. Through our unwavering dedication to scientific excellence and innovation, we strive to revolutionize the biologics industry and make a positive impact on the lives of patients in Arunachal Pradesh and beyond.
Contact Us Today:
Experience the difference with Shilpa Biologicals. For stability testing services that you can trust, contact us at +91-836 248 5555 or email us at Madhav@shilpabio.com. Partner with us and embark on a journey towards safer, more effective pharmaceutical solutions for the welfare of patients worldwide.
Leading Biologicals company in India
Shilpa Biologicals: Pioneering Biologics Solutions in India
Discover the forefront of biologics innovation with Shilpa Biologicals, India's leading company in the field. Located at Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India, Shilpa Biologicals stands as a beacon of excellence, offering cutting-edge solutions in biologics manufacturing and services.
Why Choose Shilpa Biologicals?
At Shilpa Biologicals, our mission is clear: to develop curative therapies that transform lives. With a dedicated team of scientists and professionals at our state-of-the-art R&D manufacturing facility in Dharwad, Karnataka, we focus on advancing treatments in the immunology and haematology sectors.
Unmatched Expertise and Innovation:
Our commitment to scientific excellence drives every aspect of our work. Leveraging expertise in molecular biology, biochemistry, immunology, and formulations, we have cultivated a robust pipeline of projects poised to revolutionize the biologics landscape. Our innovative solutions are designed to make life-saving treatments affordable and accessible to patients worldwide.
Global Impact, Local Presence:
While our reach extends across the globe, our roots remain firmly planted in India. We are dedicated to serving the healthcare needs of our local communities and beyond. Through strategic partnerships and collaborations, we strive to make a meaningful difference in the lives of patients everywhere.
Committed to Patient Welfare:
At Shilpa Biologicals, patient welfare is at the heart of everything we do. Our team of talented professionals is driven by a shared passion for improving healthcare outcomes. Through innovation, dedication, and unwavering commitment, we aim to set new standards of excellence in biologics manufacturing and services.
Contact Us Today:
Experience the future of biologics with Shilpa Biologicals. Call us at +91-836 248 5555 or email us at Madhav@shilpabio.com to learn more about our groundbreaking initiatives and how we can partner together for better healthcare outcomes. Join us as we pave the way for a brighter, healthier future with Shilpa Biologicals, India's premier biologics company.
Adenovirus purification in India
Looking for reliable adenovirus purification services in India? Trust Shilpa Biologicals, a renowned biologics company dedicated to advancing healthcare through innovative solutions. With our cutting-edge R&D manufacturing facility located at Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India, we offer comprehensive adenovirus purification services tailored to meet your specific needs.
Why Choose Shilpa Biologicals for Adenovirus Purification?
At Shilpa Biologicals, we specialize in biologics manufacturing and services, with a strong focus on developing curative therapies. Our dedicated team of scientists and professionals is committed to delivering high-quality adenovirus purification services that meet the stringent requirements of the biopharmaceutical industry.
State-of-the-Art Facility:
Our R&D manufacturing facility in Dharwad, Karnataka, is equipped with state-of-the-art technology and equipment, allowing us to perform adenovirus purification with precision and efficiency. We adhere to strict quality standards to ensure the purity and integrity of the final product.
Expertise in Biologics Development:
With expertise in molecular biology, biochemistry, immunology, and formulations, our team at Shilpa Biologicals is well-equipped to handle complex biologics projects, including adenovirus purification. We leverage our scientific strength and innovative approach to deliver exceptional results for our clients.
Global Reach, Local Presence:
While we operate on a global scale, our roots are firmly planted in India. We are proud to serve the healthcare needs of patients worldwide, offering affordable and accessible solutions. Whether you're located in India or abroad, you can rely on Shilpa Biologicals for high-quality adenovirus purification services.
Contact Us Today:
Experience the difference with Shilpa Biologicals. Call us at +91-836 248 5555 or email us at Madhav@shilpabio.com to learn more about our adenovirus purification services and how we can support your biologics development needs. Partner with us and discover why Shilpa Biologicals is the trusted choice for biologics manufacturing and services in India.
autoimmune disorders manufacturers in karnataka
Leading Autoimmune Disorders Manufacturers in Karnataka: Shilpa Biologicals
Seeking top-tier autoimmune disorders manufacturers in Karnataka? Look no further than Shilpa Biologicals, a pioneering biologics company dedicated to transforming healthcare through innovative solutions. Located at Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India, Shilpa Biologicals stands as a beacon of excellence in biologics manufacturing and services.
Why Choose Shilpa Biologicals for Autoimmune Disorders:
Shilpa Biologicals is committed to developing curative therapies for autoimmune disorders, leveraging our cutting-edge R&D manufacturing facility in Dharwad, Karnataka. Our team of scientists and professionals possesses the expertise and dedication needed to address the complex challenges of autoimmune diseases.
State-of-the-Art Infrastructure:
Our R&D manufacturing facility is equipped with state-of-the-art technology and equipment, enabling us to develop innovative treatments for autoimmune disorders with precision and efficiency. We adhere to stringent quality standards to ensure the safety and efficacy of our products.
Focus on Immunology:
At Shilpa Biologicals, we specialize in the immunology sector, with a focus on developing therapies for autoimmune disorders. Our product pipeline is designed to address the unmet medical needs of patients worldwide, offering hope and relief to those suffering from autoimmune diseases.
Global Impact, Local Presence:
While our reach extends globally, our roots remain firmly planted in Karnataka, India. We are proud to serve the healthcare needs of our local community and beyond, offering affordable and accessible solutions for autoimmune disorders.
Contact Us Today:
Experience the difference with Shilpa Biologicals. Call us at +91-836 248 5555 or email us at Madhav@shilpabio.com to learn more about our autoimmune disorders manufacturing services and how we can support your healthcare needs. Partner with us and discover why Shilpa Biologicals is the trusted choice for biologics manufacturing in Karnataka, India.
ShilpaBio: Innovating Biologics and CDMO Services
Located in Belur Industrial Area, Dharwad, Karnataka, ShilpaBio is at the forefront of biopharmaceutical innovation, combining expertise in molecular biology, cancer biology, and translational sciences to develop affordable biologic therapies. The company's product pipeline reflects its dedication to developing and manufacturing advanced biologics such as biosimilars, fusion proteins, and vaccines, with a particular emphasis on cell-line development, protein characterization, and formulation stability.
ShilpaBio's capabilities span from transient and stable cell line development to scalable fermentation and purification processes, with state-of-the-art analytical development, quality control labs, and R&D facilities. Their manufacturing space supports the complete cycle of drug development, ensuring high-quality production from small volume (50-200L bioreactors) to large volume (1000-2000L bioreactors). In addition, the planned expansion will elevate ShilpaBio's production capacity to 16,000 liters, further enhancing their ability to meet industry demands.
ShilpaBio excels as a Contract Development and Manufacturing Organization (CDMO), providing tailored solutions to pharmaceutical companies. Their CDMO services include:
ShilpaBio’s continuous processes for MAbs, glycoproteins, and fusion proteins guarantee production efficiency. Their integrated operations include cell banking, characterization, and filter validation, allowing partners to benefit from the company’s comprehensive biological infrastructure.
ShilpaBio's proprietary NavAlbumin Technology Platform offers novel formulation solutions, stabilizing biologic products early in the development process. This technology reduces overall costs and enhances product stability, ensuring swift market entry for new formulations.
ShilpaBio has forged strategic partnerships with global biopharmaceutical leaders. In May 2021, the company collaborated with Dr. Reddy’s Laboratories to manufacture the Sputnik vaccine, while in August 2021, they partnered with Zydus Cadila for the production of the ZyCov vaccine. These partnerships underscore ShilpaBio's role as a trusted partner in the fight against COVID-19.
ShilpaBio offers world-class packaging capabilities, including:
Their manufacturing capabilities span small to large volume production, with output ranging from 20 units/min for prefilled syringes (PFS) and vials to 300 units/min for high-volume vial filling.
ShilpaBio is committed to maintaining the highest standards of quality control, with in-house characterization and validation of processes and products. Their dedication to innovative drug discovery, cell-line development, and vaccine stabilization positions them as leaders in biologics development.
If you're interested in partnering with ShilpaBio to take your project from concept to commercialization, reach out to their Business Development team at BD_bio@shilpabio.com.
For more information on ShilpaBio’s capabilities, partnerships, and cutting-edge technologies, visit their headquarters at Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India.
Fusion protein development in India
Fusion Protein Development in India: A Growing Biopharmaceutical Frontier
India is emerging as a key player in the development of fusion proteins, a class of biologics that combine two or more proteins to create novel therapies with enhanced functionalities. These engineered proteins have shown tremendous potential in oncology, immunology, and rare diseases, providing targeted and effective treatments. India's strong biopharmaceutical infrastructure and talent pool make it an ideal hub for the research, development, and manufacturing of fusion proteins.
Despite the progress, there are challenges to overcome, including the complexity of protein design, scaling up production while maintaining product quality, and navigating the stringent global regulatory environment. However, India's growing investment in biopharma R&D, expanding biosimilar market, and focus on innovation provide vast opportunities for the development of novel fusion protein therapies.
ShilpaBio is one of the leading companies in India actively involved in fusion protein development. Their proprietary technologies, such as engineered CHO cell lines, continuous bioprocessing for glycoproteins and fusion proteins, and high-concentration liquid formulations, position them as innovators in this space. ShilpaBio's state-of-the-art facilities in Dharwad, Karnataka, and their comprehensive CDMO services provide the infrastructure needed for taking fusion protein projects from the lab to the market.
With India poised to become a global leader in biopharmaceutical innovation, the country is playing a crucial role in bringing affordable fusion protein therapies to patients worldwide.
cell line characterization in karnataka
Darbepoetin manufacturers in Gujarat
Darbepoetin Manufacturers in Gujarat: ShilpaBio - Innovators in Biologic Therapies
ShilpaBio, located in Karnataka, is a leading biopharmaceutical company with cutting-edge capabilities in the development and manufacturing of biologic therapies, including Darbepoetin. ShilpaBio's expertise spans across molecular biology, cancer biology, and translational sciences, allowing the company to create a robust pipeline of affordable biologic products. While their operations are based in Dharwad, Karnataka, they serve customers in Gujarat and other parts of India.
ShilpaBio excels in drug substance and drug product development, ensuring the highest quality standards throughout the entire manufacturing process. The company’s state-of-the-art facilities in molecular biology, biochemistry, process characterization, and scalable fermentation processes enable efficient production of Darbepoetin, a critical therapeutic used to treat anemia in patients with chronic kidney disease or those undergoing chemotherapy.
The core strength of ShilpaBio lies in its expertise in developing both transient and stable cell lines for biologics such as Darbepoetin. The company’s specialized team conducts media optimization, scalable fermentation, and purification processes in small to large bioreactors, ensuring the production of high-quality Darbepoetin. ShilpaBio’s ability to scale production from 10L to 2000L bioreactors provides flexibility and efficiency, catering to both clinical and commercial demands.
ShilpaBio’s commitment to excellence is supported by their robust Quality and Regulatory teams. They ensure compliance with international standards through rigorous validation processes. From process validation batches at clinical and commercial scales to freeze-thaw studies and degradation testing, ShilpaBio covers every aspect of Darbepoetin manufacturing. Their high-end analytical methods guarantee the complete characterization of proteins, ensuring the stability and efficacy of their biologic therapies.
ShilpaBio operates cutting-edge manufacturing facilities with both small and large volume capabilities, allowing them to cater to various customer requirements, from research to large-scale commercial manufacturing. Their facilities include:
ShilpaBio’s biologics, including Darbepoetin, are developed to meet the rising demands of the healthcare sector across India, including Gujarat. Their scalable production and efficient processes make them a reliable partner for delivering critical therapies at an affordable cost.
ShilpaBio is open to collaborations with biopharmaceutical companies and researchers to co-develop and commercialize novel biologic formulations. They have successfully partnered with leading pharmaceutical companies, such as Dr. Reddy’s Laboratories and Zydus Cadilla, for the production of COVID-19 vaccines, showcasing their capabilities as a Contract Development and Manufacturing Organization (CDMO).
For high-quality Darbepoetin manufacturing and biologic development solutions in Gujarat, partner with ShilpaBio, a trusted name in biopharmaceutical innovation. Contact us at BD_bio@shilpabio.com to explore collaborative opportunities.
ShilpaBio – Driving Innovation in Biologics Manufacturing
Monoclonal Antibody manufacturing compnay in Chhattisgarh
ShilpaBio is a pioneering company in the realm of biologics, specializing in monoclonal antibody (mAb) manufacturing. Located at Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India, ShilpaBio is dedicated to developing affordable biologic therapies by leveraging its robust expertise in molecular biology, cancer biology, and translational sciences.
Product Pipeline:
ShilpaBio’s product pipeline is built on a solid foundation of advanced research and development capabilities. The company is committed to delivering innovative drug substances and drug products, ensuring a seamless journey from concept to commercialization. The research team at ShilpaBio is composed of seasoned experts skilled in various domains, including:
This diverse expertise enables ShilpaBio to address complex challenges in drug discovery and development, positioning it as a leader in the biopharmaceutical industry.
Advanced Manufacturing Capabilities:
ShilpaBio boasts state-of-the-art facilities for drug substance (DS) and drug product (DP) manufacturing. The company’s capabilities include:
Key Capabilities:
CDMO Services:
As a Contract Development and Manufacturing Organization (CDMO), ShilpaBio provides proprietary technologies and comprehensive services to support partners in the biopharmaceutical sector. Their strengths include:
Innovation and Research:
ShilpaBio’s innovative NavAlbumin technology platform offers unique formulation strategies, enhancing product stability and reducing costs. This technology allows for rapid stabilization of products in early development, expediting the path to market.
Conclusion:
ShilpaBio is at the forefront of monoclonal antibody manufacturing in India, committed to delivering high-quality biologic therapies. With its extensive expertise, state-of-the-art facilities, and a strong focus on research and development, ShilpaBio is poised to address the evolving needs of the biopharmaceutical industry.
For inquiries or to explore partnership opportunities, please contact ShilpaBio at BD_bio@shilpabio.com.
CDMO Biologics Manufacturers in India
About Shilpa Biologicals
Shilpa Biologicals is a prominent biologics manufacturing and services company based in Dharwad, Karnataka, India. With a state-of-the-art R&D and manufacturing facility, the company focuses on delivering curative therapies in the fields of immunology and hematology. Its mission is to make advanced treatments affordable and accessible to patients worldwide.
Shilpa Biologicals leverages expertise in molecular biology, biochemistry, immunology, and formulations to develop innovative solutions. The company has established a robust pipeline of proprietary projects aimed at revolutionizing the blood-product segment and setting benchmarks in global biologics services.
Key Strengths and Vision
Key Milestones and Recent Updates
Contact Details
Shilpa Biologicals continues to drive innovation, striving to set new benchmarks in biologics manufacturing and CDMO services globally.
Albumin Characterization
Albumin characterization plays a vital role in understanding its biochemical properties, stability, and functionality for therapeutic and diagnostic applications. It is especially important in the context of biologics, where precision and quality are critical.
While Arunachal Pradesh is not traditionally recognized as a hub for advanced biologics research and manufacturing, efforts to expand such scientific capabilities in the region could involve partnerships or initiatives from leading companies like Shilpa Biologicals.
Shilpa Biologicals and Albumin Research
Shilpa Biologicals, headquartered in Dharwad, Karnataka, has significant expertise in biologics and blood-product development. The company’s robust pipeline and innovative research methods could be extended to include albumin characterization initiatives in regions like Arunachal Pradesh.
Potential areas of collaboration or focus could include:
Key Applications of Albumin Characterization
Arunachal Pradesh could benefit from advancements in this area by fostering regional expertise and contributing to the broader biologics landscape.
Shilpa Biologicals: Leading Vaccines CMO Manufacturer in India
Shilpa Biologicals, headquartered in Dharwad, Karnataka, is a key player in the field of contract manufacturing for vaccines and biologics. With a focus on innovation and affordability, the company has established itself as a trusted partner for vaccine development and production.
Expertise in Vaccine Manufacturing
Shilpa Biologicals leverages its cutting-edge R&D capabilities and robust manufacturing infrastructure to provide high-quality vaccine CMO (Contract Manufacturing Organization) services. Its strengths include:
Key Collaborations in Vaccines Manufacturing
Comprehensive CMO Services
Shilpa Biologicals offers end-to-end contract manufacturing solutions for vaccines, including:
Why Partner with Shilpa Biologicals?
Contact Information
Shilpa Biologicals continues to be at the forefront of vaccine manufacturing in India, playing a vital role in addressing global health challenges through strategic partnerships and innovative solutions.
Shilpa Biologicals: Advancing Fusion Protein Development
Shilpa Biologicals, a leading biologics manufacturing and services company based in Karnataka, has established itself as a key innovator in therapeutic protein development. While the company's primary facility is located in Dharwad, Karnataka, the opportunity for expanding capabilities to regions like Chhattisgarh could pave the way for decentralized R&D and production in fusion proteins.
Fusion Protein Development: Key Focus Areas
Fusion proteins are engineered molecules that combine two or more protein domains to create therapeutic agents with enhanced efficacy, stability, and targeted delivery. Shilpa Biologicals’ strengths in molecular biology, biochemistry, and formulations position the company as a leader in this space.
Potential Expansion to Chhattisgarh
With its strategic location and growing focus on healthcare and biotechnology, Chhattisgarh could serve as a hub for specialized R&D in fusion protein development. Shilpa Biologicals’ potential initiatives in the region might include:
Applications of Fusion Proteins
Shilpa Biologicals’ Expertise
Shilpa Biologicals combines innovation and scalability, leveraging its advanced R&D frameworks and a dedicated team of scientists to deliver high-quality biologics. The company’s expansion into fusion protein development aligns with its goal of revolutionizing therapeutic approaches and making treatments accessible worldwide.
Contact Information
Shilpa Biologicals’ expertise and forward-thinking approach could bring cutting-edge fusion protein development to Chhattisgarh, contributing to the region's scientific and healthcare landscape.
Shilpa Biologicals, a subsidiary of Shilpa Medicare Limited (SML), is a leading biologics manufacturing and services company based in Dharwad, Karnataka. Renowned for its focus on developing curative therapies, Shilpa Biologicals leverages cutting-edge scientific expertise in molecular biology, biochemistry, immunology, and formulations to create innovative solutions in the immunology and hematology sectors.
Shilpa Biologicals’ robust pipeline of wholly owned projects is designed to address unmet medical needs and transform healthcare. The company’s key focus lies in:
Shilpa Biologicals prides itself on its talented team of scientists and professionals committed to improving patient outcomes globally. By embracing innovation, the company is disrupting traditional solutions to address complex scientific challenges.
Address:
Plot 532/A, Belur Industrial Area,
Dharwad-580011, Karnataka, India
Shilpa Biologicals stands as a trusted partner in the biologics manufacturing space, striving to redefine healthcare with its innovative therapies and biosimilar products.
Shilpa Biologicals, a subsidiary of Shilpa Medicare Limited (SML), is a pioneering biologics manufacturing company with a strong focus on innovative therapies in immunology and hematology. With its R&D and manufacturing facility headquartered in Dharwad, Karnataka, Shilpa Biologicals has expanded its operations to cater to the increasing demand for high-quality biologics, including human albumin, across India, including Andhra Pradesh.
For inquiries about albumin manufacturing in Andhra Pradesh, please reach out to:
Shilpa Biologicals Private Limited
Plot 532/A, Belur Industrial Area,
Dharwad-580011, Karnataka, India
Shilpa Biologicals is your trusted partner for high-quality albumin manufacturing, ensuring excellence and reliability in every step of the production process.
Shilpa Biologicals, a subsidiary of Shilpa Medicare Limited (SML), is a leading biologics manufacturing and services company with a focus on developing advanced therapies, including fusion protein development. Based in Dharwad, Karnataka, Shilpa Biologicals is dedicated to delivering high-quality biologics to cater to the healthcare needs across India, including Chhattisgarh.
Fusion proteins are engineered molecules that combine the functional elements of two or more proteins to create a new therapeutic entity. Shilpa Biologicals leverages its expertise in molecular biology, biochemistry, and immunology to develop fusion proteins that are innovative, effective, and accessible.
Shilpa Biologicals’ innovative fusion protein therapies aim to revolutionize healthcare in Chhattisgarh by providing advanced, patient-centered solutions.
For more details on fusion protein development, connect with:
Shilpa Biologicals Private Limited
Plot 532/A, Belur Industrial Area,
Dharwad-580011, Karnataka, India
Shilpa Biologicals remains at the forefront of fusion protein development, delivering breakthrough therapies to transform healthcare outcomes in Chhattisgarh and globally.
Shilpa Biologicals, a leading biologics manufacturing and services company based in Dharwad, Karnataka, offers comprehensive stability testing services for pharmaceutical products. With a state-of-the-art R&D facility and an expert team of scientists, Shilpa Biologicals ensures high-quality stability testing solutions to meet global regulatory standards.
Stability testing is a critical process in pharmaceutical development, ensuring that a product maintains its safety, efficacy, and quality under specified environmental conditions over time. It involves evaluating how factors like temperature, humidity, and light impact the product.
Shilpa Biologicals is dedicated to advancing pharmaceutical excellence in Karnataka, providing stability testing solutions that empower clients to meet regulatory requirements and deliver high-quality products to patients worldwide.
For stability testing inquiries, connect with:
Shilpa Biologicals Private Limited
Plot 532/A, Belur Industrial Area,
Dharwad-580011, Karnataka, India
Shilpa Biologicals is your trusted partner for stability testing of pharmaceutical products, ensuring innovative and reliable healthcare solutions.
Shilpa Biologicals, based in Dharwad, Karnataka, is a premier Contract Development and Manufacturing Organization (CDMO) specializing in biologics manufacturing and services. Our state-of-the-art R&D and manufacturing facility is dedicated to developing curative therapies that can positively impact patients worldwide. We focus on providing high-quality services in immunology and hematology, leveraging cutting-edge scientific advancements to create affordable, life-saving treatments.
At Shilpa Biologicals, we take pride in our robust pipeline of biologic products and services, built on the strong foundations of molecular biology, biochemistry, immunology, and formulations. Our talented team of scientists and professionals is committed to making a meaningful difference in the global healthcare landscape by pioneering breakthrough therapies. We are particularly focused on revolutionizing the blood-product segment and offering world-class services that meet the highest scientific standards.
As one of India's leading biologics manufacturers, we offer a broad spectrum of CDMO services, from early-stage development to large-scale commercial manufacturing. Our goal is to provide end-to-end solutions that enable our clients to bring life-changing biologics to market with speed, precision, and regulatory compliance.
We are proud to introduce ORIADALI (Adalimumab 40 mg/0.4 mL), a cutting-edge solution for the treatment of autoimmune diseases. This pre-filled syringe, designed for subcutaneous use, is manufactured with the highest standards of quality, ensuring that patients receive precision care. With ORIADALI, we aim to provide reliable and affordable treatments, empowering patients worldwide to take control of their health.
Our leadership team at Shilpa Biologicals comprises visionary professionals committed to advancing the company’s mission and driving scientific innovation. Meet the leaders who are paving the way for ShilpaBio’s growth and success:
Our leadership team brings together a wealth of expertise and experience, helping us stay at the forefront of the biologics manufacturing industry.
Shilpa Biologicals is proud to offer a diverse product pipeline, with a focus on biosimilars, CDMO services, and strategic partnerships. We are constantly expanding our portfolio to address unmet medical needs in immunology and hematology, ensuring that patients across the globe have access to safe, effective, and affordable therapies.
As a trusted CDMO partner, Shilpa Biologicals provides comprehensive solutions to pharmaceutical and biotechnology companies. Our capabilities include:
We are committed to delivering top-notch services, fostering long-term partnerships, and advancing global healthcare through the power of biologics.
To learn more about Shilpa Biologicals and our cutting-edge biologics manufacturing services, please contact us:
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India
Phone: +91-836 248 5555
Email: madhav@vbshilpa.com
At Shilpa Biologicals, we are at the heart of scientific innovation, revolutionizing biologics manufacturing and improving lives worldwide. Partner with us for groundbreaking solutions that make a difference.
Shilpa Biologicals, a leading biologics manufacturing and services company, offers innovative fusion protein manufacturing solutions from our cutting-edge R&D and manufacturing facility in Dharwad, Karnataka. While we are based in Karnataka, we have the expertise to serve clients in Andhra Pradesh and across India. With a strong commitment to advancing curative therapies, our team of world-class scientists and professionals works tirelessly to provide groundbreaking biological solutions that transform healthcare and improve the lives of patients worldwide.
Fusion proteins are a powerful class of biologics with applications in immunology, hematology, and oncology. At Shilpa Biologicals, we specialize in the development and manufacturing of high-quality fusion proteins that can effectively target disease mechanisms and provide life-saving treatments. We leverage our expertise in molecular biology, biochemistry, immunology, and formulations to produce fusion proteins with precision and reliability.
Our R&D facility is equipped with the latest technologies and infrastructure to support the entire fusion protein development lifecycle, from early-stage research to large-scale commercial production. We aim to make innovative biologics, including fusion proteins, accessible and affordable for patients globally.
At Shilpa Biologicals, our robust product pipeline focuses on addressing critical medical needs in the fields of immunology and hematology. Our fusion protein manufacturing capabilities are part of our larger mission to deliver affordable and effective biologics to patients worldwide. In addition to fusion proteins, our product portfolio includes biosimilars and other biologic therapies that promise to disrupt the status quo in medical treatments.
The leadership team at Shilpa Biologicals is committed to advancing the company's vision of revolutionizing the biologics sector. Our leaders bring years of experience and expertise to guide the company's progress:
Our leadership is focused on achieving excellence in biologics manufacturing, including the production of fusion proteins, and ensuring the highest quality standards for the global market.
As part of our commitment to scientific advancement, we are proud to introduce ORIADALI, an advanced Adalimumab solution for injection. This pre-filled syringe (40 mg/0.4 mL) is designed for subcutaneous use and is manufactured with precision and reliability by Shilpa Biosciences. ORIADALI is a testament to our capabilities in biologics manufacturing, offering targeted treatment options for autoimmune diseases and ensuring affordable and accessible care for patients globally.
As a trusted CDMO partner, Shilpa Biologicals provides a wide range of services to support the development and production of fusion proteins and other biologic therapies. Our CDMO capabilities include:
If you are seeking a reliable fusion protein manufacturer in Andhra Pradesh or elsewhere in India, Shilpa Biologicals is your ideal partner. We are committed to delivering high-quality biologics that help address pressing healthcare needs.
For more information or to discuss a potential partnership, please contact us:
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India
Phone: +91-836 248 5555
Email: madhav@vbshilpa.com
Shilpa Biologicals is at the forefront of fusion protein manufacturing, providing innovative and affordable solutions to help transform global healthcare. Let us help you bring your biologics to life with our cutting-edge manufacturing capabilities.
Shilpa Biologicals, a leading biologics manufacturing and services company, is focused on the development of advanced glycoproteins and other biologic therapies. While our state-of-the-art R&D and manufacturing facility is located in Dharwad, Karnataka, we proudly extend our expertise to Chhattisgarh and other regions across India, helping to revolutionize the biopharmaceutical landscape.
At Shilpa Biologicals, we are committed to advancing the science of glycoprotein development, leveraging the latest advancements in molecular biology, biochemistry, and immunology. Our cutting-edge team of scientists is dedicated to developing curative therapies that offer affordable solutions for patients worldwide.
Glycoproteins play a critical role in a wide range of biological processes, including immune response, cell signaling, and enzymatic activity. As a pioneer in biologics manufacturing, Shilpa Biologicals focuses on the precise development of high-quality glycoproteins that are essential for creating effective therapies in immunology and hematology.
Our scientific strength in glycoprotein development enables us to optimize glycosylation processes and improve the efficacy and stability of therapeutic proteins. We are committed to producing glycoproteins that meet the highest standards of quality, ensuring both reliability and safety for patients worldwide.
Shilpa Biologicals combines deep scientific knowledge with an innovative approach to produce glycoproteins for advanced biologic therapies. With our expertise in biochemistry and formulations, we continuously optimize production processes to improve the yield, quality, and cost-effectiveness of glycoprotein production.
Our product pipeline in the immunology and hematology sectors includes glycoprotein-based therapies designed to address critical medical needs. Through our strong commitment to research and development, we aim to create treatments that are accessible and affordable, benefiting patients globally.
Shilpa Biologicals is proud to introduce ORIADALI, an advanced Adalimumab solution for injection, designed for precision care in the treatment of autoimmune diseases. Manufactured with the highest quality standards, ORIADALI (40 mg/0.4 mL) is a pre-filled syringe that ensures reliable and effective treatment for patients. Our focus on innovative glycoprotein development plays a key role in ensuring the success of biologic treatments like ORIADALI.
Shilpa Biologicals is driven by a team of experienced and visionary leaders who guide the company’s progress and growth in the biologics sector. Meet the leadership team behind our glycoprotein and biologics development efforts:
Our leadership is dedicated to driving the mission of Shilpa Biologicals forward and expanding our capabilities in the glycoprotein and biologics sectors.
Shilpa Biologicals’ robust product pipeline encompasses biosimilars, glycoproteins, and other advanced biologic therapies aimed at addressing global healthcare challenges. We are committed to advancing treatments in immunology and hematology, making cutting-edge therapies more accessible and affordable to patients in India and around the world.
As a leading CDMO, Shilpa Biologicals offers a range of services to help develop and manufacture glycoproteins and other biologics:
If you are looking for expert glycoprotein development services in Chhattisgarh or across India, Shilpa Biologicals is your trusted partner. We are committed to advancing biologic therapies and providing solutions that benefit patients worldwide.
To learn more about our glycoprotein development services, please contact us:
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India
Phone: +91-836 248 5555
Email: madhav@vbshilpa.com
Shilpa Biologicals is at the forefront of glycoprotein development, offering cutting-edge biologics manufacturing services. Partner with us to bring advanced biologic therapies to market, improving the lives of patients worldwide.
Shilpa Biologicals is a leading biologics manufacturing and services company, offering comprehensive Contract Manufacturing Organization (CMO) services for vaccine production. While our cutting-edge R&D and manufacturing facility is based in Dharwad, Karnataka, we are proud to extend our high-quality services to Bihar and other regions across India. Our focus is on developing and manufacturing innovative vaccines and biologics that address critical healthcare needs globally.
At Shilpa Biologicals, we specialize in providing top-tier vaccines CMO services for the pharmaceutical and biotechnology sectors. We support our clients with end-to-end vaccine development and manufacturing solutions. From early-stage development to large-scale commercial production, our expertise in molecular biology, immunology, and biochemistry ensures the highest quality vaccines for addressing a range of infectious diseases and other global health challenges.
Our state-of-the-art manufacturing facility in Dharwad, Karnataka, supports vaccine production in line with international standards. We are committed to delivering vaccines that are not only scientifically advanced but also accessible and affordable for patients in Bihar and across the globe.
Shilpa Biologicals’ leadership team plays an instrumental role in the company’s success. They guide the organization’s commitment to high-quality biologic and vaccine manufacturing:
Shilpa Biologicals has a diverse product pipeline, including biosimilars, vaccines, and other biologic therapies. We focus on immunology and hematology sectors to address unmet medical needs. Our CDMO services allow us to partner with pharmaceutical companies and biotech firms to develop and manufacture vaccines with the highest scientific rigor and production quality.
While ORIADALI is not a vaccine, it represents Shilpa Biologicals’ commitment to innovation in biologics. ORIADALI is an advanced Adalimumab solution designed for subcutaneous use, manufactured to the highest standards of quality. This biologic therapy showcases our dedication to producing safe and effective biologics for critical healthcare needs.
Shilpa Biologicals offers an array of CMO services that make us a trusted partner for vaccine manufacturing:
If you're looking for a reliable vaccines CMO manufacturer in Bihar or across India, Shilpa Biologicals offers high-quality vaccine manufacturing services to help you bring your vaccine solutions to market. Our expertise, cutting-edge facilities, and commitment to excellence make us the ideal partner for vaccine production.
For more information on our CMO services or to discuss a potential collaboration, please contact us:
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India
Phone: +91-836 248 5555
Email: madhav@vbshilpa.com
Shilpa Biologicals is committed to advancing healthcare with vaccine manufacturing and biologics services that meet the highest international standards. Partner with us to bring innovative vaccines to patients in Bihar and across the world.
CDMO Biologics Manufacturers in India
Shilpa Biologicals
Shilpa Biologicals, based in Dharwad, Karnataka, is a leading Contract Development and Manufacturing Organization (CDMO) specializing in biologics manufacturing and services. With a focus on cutting-edge curative therapies, Shilpa Biologicals is committed to offering high-quality products in the immunology and hematology sectors, aiming to make life-changing therapies affordable and accessible to patients worldwide.
Innovative Solutions and Expertise in Biologics
At Shilpa Biologicals, we leverage our deep scientific expertise in molecular biology, biochemistry, immunology, and formulations to develop groundbreaking biologics that have the potential to disrupt the global blood-product segment. With a dedicated team of scientists and professionals at the forefront of innovation, we are well-positioned to compete globally for best-in-class services in biologics manufacturing.
Our robust pipeline of wholly owned projects is designed to address significant scientific challenges and introduce new-age solutions to the biologics industry. The focus is on creating affordable and effective therapies that benefit patients around the world.
ORIADALI: Advanced Adalimumab Solution for Injection
One of our flagship products, ORIADALI (Adalimumab 40 mg/0.4 mL), is a cutting-edge, pre-filled syringe designed for subcutaneous use. Manufactured with the highest standards of quality and precision, ORIADALI provides reliable care for patients, ensuring they receive effective treatment. This product exemplifies our commitment to innovation and high-quality standards in the biologics industry.
Leadership Team at Shilpa Biologicals
Shilpa Biologicals' success is driven by a talented and visionary leadership team:
Our leadership is committed to fostering innovation and achieving the full potential of Shilpa Biologicals in the biologics sector.
CDMO Services & Capabilities
As a CDMO, Shilpa Biologicals offers a wide range of services, from product development to large-scale manufacturing. Our capabilities include:
We are trusted by global partners to deliver high-quality biologics and services tailored to their specific needs. Our facilities are equipped with state-of-the-art technology to ensure precision and excellence in every stage of production.
Global Partnerships & Collaborations
We are committed to fostering strategic partnerships and collaborations with global players in the biologics and pharmaceutical industries. Our partnerships aim to bring innovative therapies to market faster, ensuring patients worldwide benefit from life-saving treatments.
Contact Shilpa Biologicals
For more information on our products, services, and partnerships, contact us at:
Conclusion
Shilpa Biologicals is dedicated to being at the forefront of biologics innovation in India. With a focus on biosimilars, CDMO services, and a robust product pipeline, we are leading the way in the development and manufacture of biologic therapies that address global health challenges. Our scientific expertise, state-of-the-art facilities, and leadership in biologics manufacturing make us a trusted partner for businesses and patients alike.
Monoclonal Antibody manufacturing compnay in Andhra Pradesh
Shilpa Biologicals, a leading biologics manufacturing and services company, is committed to advancing the development of curative therapies. While our primary R&D and manufacturing facility is based in Dharwad, Karnataka, we are rapidly expanding our footprint to support clients across India, including those in Andhra Pradesh, with high-quality monoclonal antibody (mAb) manufacturing and services.
Innovative Solutions in Monoclonal Antibody Manufacturing
At Shilpa Biologicals, we focus on creating cutting-edge therapies, with a particular emphasis on monoclonal antibodies (mAbs) in immunology and hematology. Our team of talented scientists and professionals is dedicated to using the latest technologies and methodologies to create therapies that make a significant impact on patient health. We leverage our expertise in molecular biology, biochemistry, immunology, and formulations to develop a robust pipeline of innovative products.
A World-Class Monoclonal Antibody Manufacturing Facility
Our advanced R&D facility, while located in Karnataka, serves as a key hub for monoclonal antibody research and manufacturing. By continuously improving our processes, we offer cutting-edge, scalable solutions for the development and commercial-scale production of monoclonal antibodies. Our dedication to quality ensures that every monoclonal antibody product we manufacture meets the highest industry standards.
A Focus on Affordability and Global Reach
Shilpa Biologicals is committed to developing biologics, including monoclonal antibodies, that are both scientifically advanced and affordable for patients worldwide. With a strong pipeline that focuses on immunology and hematology, we are well-positioned to address global healthcare needs and provide solutions that revolutionize the treatment of chronic and complex diseases.
Product Highlight: ORIADALI - Advanced Adalimumab Solution for Injection
An example of our cutting-edge work in biologics is ORIADALI (Adalimumab 40 mg/0.4 mL), a pre-filled syringe designed for subcutaneous use. Manufactured by Shilpa Biosciences, ORIADALI is an advanced solution for patients requiring adalimumab, offering precision care, reliability, and the highest standards in biologics treatment. This product exemplifies our commitment to providing affordable and accessible therapies globally.
Leadership at Shilpa Biologicals
The success of Shilpa Biologicals is powered by our visionary leadership team, which includes:
Our leadership’s experience and dedication drive the company's mission to provide innovative solutions for the betterment of patients worldwide.
Our Services and Capabilities in Monoclonal Antibody Manufacturing
Shilpa Biologicals offers comprehensive services in monoclonal antibody manufacturing, including:
We provide end-to-end services to our global clients, ensuring the seamless transition from discovery to commercial-scale production. Our facility is equipped with state-of-the-art technology to produce monoclonal antibodies that meet stringent regulatory requirements and patient needs.
Partnerships and Global Collaborations
Shilpa Biologicals actively seeks collaborations with companies and organizations in Andhra Pradesh and across India. By partnering with key industry players, we aim to bring innovative therapies to market quickly, ensuring that patients worldwide have access to life-changing treatments.
Contact Shilpa Biologicals for Monoclonal Antibody Manufacturing
For more information on our monoclonal antibody manufacturing services, product offerings, or partnership opportunities, please contact us:
Conclusion
Shilpa Biologicals stands at the forefront of biologics manufacturing in India, including the production of monoclonal antibodies. With our scientific strength, commitment to quality, and leadership in R&D, we are proud to offer innovative and affordable therapies to patients globally. Our focus on immunology, hematology, and the continuous development of biologic therapies underscores our mission to make a meaningful impact on global health. As we expand our services, we are excited to collaborate with partners in Andhra Pradesh and across the world to revolutionize the biologics landscape.
Stability Testing OfPharmaceutical Products in Arunachal Pradesh
Shilpa Biologicals, a renowned biologics manufacturing and services company, specializes in the development of curative therapies with a focus on immunology and hematology. While our primary R&D and manufacturing facility is based in Dharwad, Karnataka, our commitment to advancing pharmaceutical solutions, including stability testing, extends to all regions, including Arunachal Pradesh.
Importance of Stability Testing in Pharmaceuticals
Stability testing is a critical component in the pharmaceutical industry to ensure the safety, efficacy, and quality of pharmaceutical products throughout their shelf life. At Shilpa Biologicals, we prioritize stability testing to ensure that all our biologic products, including monoclonal antibodies and biosimilars, maintain their effectiveness and safety under various environmental conditions.
Our stability testing protocols are designed to assess factors such as temperature, humidity, and light exposure, helping ensure that our products remain safe and effective for patients worldwide. This testing is essential for regulatory approvals and to meet industry standards, ensuring that every product we manufacture is of the highest quality.
Shilpa Biologicals' Expertise in Stability Testing
Shilpa Biologicals has built a reputation for scientific strength and a commitment to high-level breakthroughs in the biologics industry. Our experienced scientists and professionals utilize cutting-edge technologies and methodologies to conduct thorough stability testing on our product pipeline, which focuses on immunology, hematology, and biologics manufacturing.
By applying our deep knowledge in molecular biology, biochemistry, immunology, and formulations, we conduct comprehensive stability studies to ensure our products remain stable under a wide range of conditions. Our testing process is part of our commitment to global healthcare standards, ensuring that every product we develop, including those destined for the market in Arunachal Pradesh, is safe, effective, and reliable.
Advanced Product Solutions: ORIADALI
One example of our commitment to product quality is ORIADALI (Adalimumab 40 mg/0.4 mL), a pre-filled syringe for subcutaneous use. Manufactured by Shilpa Biosciences, ORIADALI undergoes rigorous stability testing to ensure it maintains its potency and effectiveness throughout its shelf life. This product exemplifies our dedication to providing patients with high-quality biologic therapies that are stable and reliable.
Leadership at Shilpa Biologicals
Our leadership team, composed of visionary professionals, drives Shilpa Biologicals' commitment to excellence in every aspect of pharmaceutical manufacturing, including stability testing:
These leaders guide our organization in delivering top-tier biologic therapies while maintaining the highest standards in stability testing and other key areas of pharmaceutical manufacturing.
Our Comprehensive Stability Testing Services
Shilpa Biologicals offers a wide range of services related to stability testing, ensuring that every product we manufacture is of the highest quality:
These services are integral to our CDMO offerings, which include the development and manufacturing of biologics such as monoclonal antibodies, biosimilars, and more.
Global Reach & Local Commitment in Arunachal Pradesh
Although our primary facility is located in Karnataka, Shilpa Biologicals' reach extends across India, including Arunachal Pradesh. We are dedicated to ensuring that our products and services meet the needs of our clients in all regions of India, including those seeking high-quality stability testing for pharmaceutical products.
Contact Shilpa Biologicals for Stability Testing Services
For more information on stability testing services or to learn about our biologics manufacturing capabilities, please contact us:
Conclusion
Shilpa Biologicals is committed to the rigorous and precise testing of pharmaceutical products, including stability studies, to ensure that our biologics meet global quality standards. By offering advanced stability testing, we guarantee that our products, such as monoclonal antibodies and biosimilars, remain safe and effective for patients. Our dedication to quality and scientific innovation makes us a trusted partner in the pharmaceutical industry, with a focus on meeting the needs of regions like Arunachal Pradesh.
Albumin Development Manufacturers in Chhattisgarh: Shilpa Biologicals
Shilpa Biologicals, a leading biologics manufacturing and services company, specializes in the development and manufacturing of innovative therapies, including albumin products. With our R&D and manufacturing facility based in Dharwad, Karnataka, we have extended our capabilities to meet the growing demand for biologics and albumin therapies in India, including Chhattisgarh.
Innovative Albumin Development at Shilpa Biologicals
At Shilpa Biologicals, we are committed to developing high-quality albumin products for therapeutic applications. Our focus on albumin development is backed by our strong expertise in molecular biology, biochemistry, immunology, and formulations. We utilize cutting-edge technologies and the latest scientific breakthroughs to manufacture albumin products that meet global standards and ensure patient safety and efficacy.
Albumin, a key protein in the human blood, is essential for a variety of medical treatments, including liver disease, burns, and surgery recovery. Shilpa Biologicals is focused on making these therapies affordable and accessible to patients across the globe, including in regions like Chhattisgarh, where there is a growing need for high-quality biologics.
Shilpa Biologicals' Expertise in Biologics Manufacturing
Our product pipeline is diverse and focuses on immunology and hematology, with a special emphasis on blood-derived products like albumin. Leveraging our deep scientific knowledge, we have developed a robust pipeline of wholly owned projects that have the potential to revolutionize the blood-product segment. Our state-of-the-art manufacturing facility is designed to ensure that all our products, including albumin, meet the highest standards of quality, safety, and effectiveness.
Why Choose Shilpa Biologicals for Albumin Development?
Introducing ORIADALI: Advanced Adalimumab Solution for Injection
In addition to albumin products, Shilpa Biologicals also develops and manufactures a range of biologics, including ORIADALI (Adalimumab 40 mg/0.4 mL), a pre-filled syringe designed for subcutaneous use. This innovative biologic therapy reflects our commitment to quality and precision care. Manufactured by Shilpa Biosciences, ORIADALI guarantees reliability in treatment, ensuring the best care for patients worldwide.
Leadership at Shilpa Biologicals
Shilpa Biologicals is driven by an experienced and visionary leadership team:
Their leadership ensures that Shilpa Biologicals remains at the forefront of biologics innovation, delivering groundbreaking therapies to patients globally.
Our Services: CDMO and Regulatory Expertise
In addition to albumin development, Shilpa Biologicals offers Contract Development and Manufacturing Organization (CDMO) services for a wide range of biologic products. Our services include:
Our CDMO services support global clients in bringing high-quality biologics to market, including albumin and other critical therapies.
Partnerships and Global Reach
Shilpa Biologicals is actively seeking strategic partnerships and collaborations with organizations and healthcare providers in Chhattisgarh and across India. By partnering with key players in the industry, we aim to accelerate the availability of albumin therapies and other life-saving biologics to patients in need.
Contact Shilpa Biologicals for Albumin Development Services
For more information on our albumin development services or to discuss potential partnerships, contact us:
Conclusion
Shilpa Biologicals is at the forefront of biologics innovation in India, offering high-quality albumin development and manufacturing services. With a focus on affordability, safety, and global standards, we are committed to delivering therapeutic solutions that make a significant impact on patient care. Our leadership in biologics manufacturing ensures that patients in Chhattisgarh and around the world have access to life-saving therapies like albumin and other biologic treatments. We look forward to partnering with healthcare providers and organizations to meet the growing need for biologics in India.
CDMO Biologics Manufacturers in Arunachal Pradesh – Shilpa Biologicals
If you're searching for reliable CDMO biologics manufacturers in Arunachal Pradesh, Shilpa Biologicals stands out as a trusted name in the biologics contract development and manufacturing space. Though our state-of-the-art R&D and manufacturing facility is located in Dharwad, Karnataka, our impact and partnerships extend across India, including the North-Eastern states like Arunachal Pradesh.
Shilpa Biologicals is a leading biologics manufacturing and services company with a strong focus on immunology and hematology. Backed by a cutting-edge team of scientists, we develop and manufacture curative therapies that are not only innovative but also accessible and affordable to patients across the globe.
We specialize in CDMO services rooted in our deep scientific capabilities in molecular biology, biochemistry, immunology, and pharmaceutical formulations. Our robust product pipeline includes breakthrough biologics designed to revolutionize treatment in critical therapeutic areas.
Experience precision biologics with ORIADALI, our advanced Adalimumab 40 mg/0.4 mL solution in a pre-filled syringe for subcutaneous administration. Manufactured by Shilpa Biosciences, ORIADALI ensures high-quality, reliable treatment for inflammatory conditions and autoimmune disorders.
We provide comprehensive CDMO biologics services tailored to the needs of biotech and pharma companies in Arunachal Pradesh and beyond:
Our scientific excellence and regulatory compliance ensure that your product development journey is seamless from concept to commercialization.
Plot 532/A, Belur Industrial Area, Dharwad – 580011, Karnataka, India
Call Us: +91-836-248-5555
Email: madhav@vbshilpa.com
Whether you’re a startup, established pharma company, or research institute in Arunachal Pradesh looking to partner with a top-tier CDMO biologics manufacturer, Shilpa Biologicals is your ideal choice. Let’s innovate together for a healthier tomorrow.
Bevacizumab manufacturers in Assam
Looking for trusted Bevacizumab manufacturers in Assam? Shilpa Biologicals is a leading name in the development and manufacturing of high-quality biologics, including biosimilars like Bevacizumab. With a state-of-the-art R&D and manufacturing facility in Dharwad, Karnataka, our reach extends across India, including the healthcare hubs of Assam and the North-East region.
Shilpa Biologicals is a cutting-edge biologics manufacturing and CDMO services company focused on delivering curative therapies in the areas of immunology and haematology. Our mission is to make innovative biologic therapies, like Bevacizumab, affordable and accessible to patients worldwide.
Our strength lies in advanced scientific capabilities across molecular biology, biochemistry, immunology, and formulations, enabling us to develop a robust product pipeline that includes biosimilars, targeted therapies, and injectables.
Bevacizumab is a vital monoclonal antibody used in the treatment of various cancers by inhibiting angiogenesis (formation of new blood vessels). At Shilpa Biologicals, our biosimilar version of Bevacizumab is manufactured under stringent quality control, ensuring global compliance, therapeutic efficacy, and cost-effectiveness—a significant step forward for oncology treatment in regions like Assam.
Though headquartered in Karnataka, we are proud to support healthcare initiatives, hospitals, and distributors in Assam by offering world-class biologic manufacturing and biosimilar solutions. Shilpa Biologicals is your reliable partner in expanding access to high-quality, life-saving therapies.
Shilpa Biologicals Pvt. Ltd.
Plot No. 532/A, Belur Industrial Area, Dharwad – 580011, Karnataka, India
Phone: +91-836-248-5555
Email: madhav@vbshilpa.com
Shilpa Biologicals – Enabling Access to Advanced Biosimilars Like Bevacizumab in Assam
Empowering patients. Advancing science. Delivering hope.
Biosimilars Manufacturers in Bihar
Are you searching for biosimilars manufacturers in Bihar? Look no further than Shilpa Biologicals, a pioneering biologics manufacturing and services company dedicated to developing affordable and advanced biosimilar therapies for global healthcare. Though headquartered in Dharwad, Karnataka, our reach and impact extend to medical institutions, hospitals, and healthcare providers across Bihar, enabling access to world-class biologics.
At Shilpa Biologicals, we are committed to transforming the treatment landscape in immunology and haematology by delivering biosimilars that are both high-quality and cost-effective. Our R&D and manufacturing facility is powered by a team of expert scientists and passionate professionals who are working on a robust pipeline of biosimilars, aiming to make breakthrough therapies more accessible to patients everywhere, including in Bihar.
Our deep expertise in molecular biology, immunology, biochemistry, and formulations has enabled us to emerge as one of the most reliable and future-ready biosimilar manufacturers in India.
We are continuously expanding our biosimilar offerings, including:
These biosimilars are developed under stringent regulatory standards, ensuring global quality and affordability, especially for the evolving healthcare needs of regions like Bihar.
Our vision is steered by a strong executive team including:
Together, they lead the charge in making Shilpa Biologicals a name trusted across India, including in underserved healthcare regions such as Bihar.
Whether you're a hospital, clinic, or distributor in Bihar looking for reliable biosimilar manufacturers, Shilpa Biologicals is your strategic partner for quality, affordability, and innovation in healthcare.
Shilpa Biologicals Pvt. Ltd.
Plot No. 532/A, Belur Industrial Area, Dharwad – 580011, Karnataka, India
Phone: +91-836-248-5555
Email: madhav@vbshilpa.com
Shilpa Biologicals – Advancing Access to Biosimilars in Bihar
Empowering treatment. Elevating care. Enhancing lives.
Erythropoetin manufacturers in India
Looking for trusted Erythropoietin manufacturers in India? Shilpa Biologicals, headquartered in Dharwad, Karnataka, stands at the forefront of biologics innovation and manufacturing. Our mission is to deliver affordable, high-quality biologics and biosimilars, including advanced Erythropoietin therapies, that meet global standards and improve patient outcomes across India and the world.
As a leading biologics manufacturing and services company, Shilpa Biologicals focuses on cutting-edge research and curative therapy development. Our R&D and manufacturing facility in Dharwad is equipped with advanced infrastructure and operated by a talented team of scientists, regulatory experts, and production professionals who are committed to delivering life-changing therapies in the fields of immunology and haematology.
Erythropoietin (EPO) is a critical biologic used to treat anemia, especially in patients with chronic kidney disease, cancer, or those undergoing dialysis. At Shilpa Biologicals, we are proud to develop and manufacture biosimilar Erythropoietin with precision, efficacy, and safety. Our EPO products are designed to meet the therapeutic needs of both clinicians and patients by offering:
Whether you're a healthcare provider, hospital, or distributor looking for Erythropoietin suppliers in India, our solutions ensure consistent therapeutic performance and affordability.
Together, they guide Shilpa Biologicals to be a global leader in biosimilars and therapeutic biologics.
In addition to Erythropoietin, we manufacture several key biosimilars, such as:
Our products are developed to revolutionize the blood-product segment and make a meaningful impact in areas with unmet medical needs.
As a trusted name among biosimilar manufacturers in India, Shilpa Biologicals ensures excellence in production, transparent processes, and a patient-first approach. Whether you are in the healthcare sector or part of the pharma supply chain, we welcome collaboration opportunities across India.
Shilpa Biologicals Pvt. Ltd.
Plot No. 532/A, Belur Industrial Area, Dharwad – 580011, Karnataka, India
Phone: +91-836-248-5555
Email: madhav@vbshilpa.com
Shilpa Biologicals – Leading Erythropoietin Manufacturer in India
Delivering Affordable Biologics. Empowering Better Health.
Shilpa Biologicals, headquartered in Dharwad, Karnataka, stands out as a premier manufacturer of Bevacizumab in India, alongside a growing portfolio of biosimilars and biologics. As a division focused on research, development, and manufacturing of biologics, Shilpa Biologicals is committed to revolutionizing patient care through cutting-edge scientific innovation and high-quality therapeutic solutions.
Our state-of-the-art R&D and manufacturing facility in Belur Industrial Area, Dharwad, is driven by a team of highly skilled scientists and professionals. Specializing in immunology and haematology, our focus is to make advanced, life-saving treatments like Bevacizumab affordable and accessible to patients across the globe.
Shilpa Biologicals’ strength lies in deep scientific expertise across molecular biology, biochemistry, immunology, and advanced formulations, enabling us to develop biosimilars and curative therapies with global competitiveness.
Our biosimilar product pipeline reflects our mission to challenge global standards and meet critical healthcare needs. One of our latest innovations includes:
Shilpa Biologicals is also actively expanding its biosimilar range to include Bevacizumab, used widely in cancer treatment, particularly in therapies targeting colorectal, lung, and renal cancers.
In addition to in-house products, Shilpa Biologicals offers CDMO (Contract Development and Manufacturing Organization) services, empowering biotech partners to scale and succeed. With robust capabilities across process development, analytical services, and regulatory support, we help partners from early-stage development through commercial manufacturing.
Together, they lead with vision, pushing the boundaries of what’s possible in biologics and biosimilar manufacturing.
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
Call Us: +91-836 248 5555
Email: madhav@vbshilpa.com
Are you looking for pharmaceutical stability testing services in Goa? Shilpa Biologicals, a premier biologics manufacturing and CDMO company based in Dharwad, Karnataka, offers advanced and reliable stability testing for pharmaceutical products, including biologics, biosimilars, and injectables.
Our state-of-the-art R&D and manufacturing facility, backed by deep scientific expertise and regulatory knowledge, supports pharmaceutical companies in Goa and across India with end-to-end analytical solutions that ensure drug product integrity, efficacy, and shelf-life compliance.
Stability testing is a critical step in the drug development lifecycle. It determines how environmental factors such as temperature, humidity, and light impact a pharmaceutical product over time. At Shilpa Biologicals, our stability studies are conducted in ICH-compliant conditions using validated methods to assess:
Whether you're developing a biologic, biosimilar, injectable, or blood product, our stability testing capabilities are designed to meet both domestic and international regulatory standards.
Though located in Dharwad, Karnataka, Shilpa Biologicals extends its analytical and stability testing services to pharma companies in Goa, a hub of pharmaceutical manufacturing in India. We understand the high standards required by regulatory bodies and offer customized testing protocols suited to your product and development phase.
With a strong product pipeline in immunology and haematology, Shilpa Biologicals is redefining patient care through innovative therapies. Our scientists combine strengths in molecular biology, biochemistry, immunology, and advanced formulations to support every stage of pharmaceutical development — from concept to commercialization.
We proudly manufacture ORIADALI, a biosimilar of Adalimumab (40 mg/0.4 mL), delivered via pre-filled syringe for subcutaneous use. It reflects our commitment to precision care, regulatory excellence, and patient-first innovation.
Our leadership and scientific teams are driving the future of healthcare innovation with passion and integrity.
Facility Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
Phone: +91-836 248 5555
Email: madhav@vbshilpa.com
Searching for reliable MSAT (Manufacturing, Scale-Up, and Technology Transfer) manufacturers in Goa? Shilpa Biologicals, headquartered in Dharwad, Karnataka, is a leading biologics manufacturing and services company offering world-class MSAT capabilities. Our scientific expertise and cutting-edge manufacturing facility support pharma and biotech companies in Goa and beyond to streamline their scale-up and tech transfer processes with precision and quality.
At Shilpa Biologicals, our dedicated team of scientists and manufacturing professionals leverage deep experience in molecular biology, biochemistry, immunology, and pharmaceutical formulations to deliver robust MSAT services that:
With a strong focus on immunology and haematology, our product pipeline and manufacturing expertise make us a preferred MSAT partner for companies aiming to bring affordable, high-quality biologics to market.
Based out of our state-of-the-art R&D and manufacturing facility in Dharwad, we combine advanced scientific methodologies with scalable production capabilities. Our innovative approach drives efficiency, reduces time-to-market, and mitigates risks during manufacturing scale-up and transfer stages.
As a testament to our scientific excellence, we proudly present ORIADALI (Adalimumab 40 mg/0.4 mL), a pre-filled syringe biosimilar designed for subcutaneous injection. Manufactured under stringent quality control, ORIADALI exemplifies our ability to deliver reliable, patient-friendly biologic therapies.
Our visionary leaders drive innovation and quality across every stage of manufacturing and development.
Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
+91-836 248 5555
madhav@vbshilpa.com
Looking for a trusted manufacturer of therapies for autoimmune disorders in Gujarat? Shilpa Biologicals, with its advanced R&D and manufacturing facility based in Dharwad, Karnataka, is a leading biologics manufacturing and services company focused on developing curative therapies for immunology and haematology sectors.
Our strong scientific foundation and cutting-edge manufacturing capabilities position us as a preferred partner for pharmaceutical companies in Gujarat seeking affordable and high-quality biologics for autoimmune conditions.
Autoimmune diseases require precision-engineered biologics for effective treatment. Shilpa Biologicals leverages expertise in molecular biology, biochemistry, immunology, and drug formulation to create innovative biosimilars and curative therapies that aim to improve patient outcomes globally.
Our portfolio includes biosimilars like ORIADALI (Adalimumab 40 mg/0.4 mL) — an advanced pre-filled syringe solution designed for subcutaneous use, ideal for treating autoimmune conditions such as rheumatoid arthritis, psoriasis, and Crohn’s disease.
Our mission is to make breakthrough therapies accessible and affordable to patients in Gujarat and worldwide, ensuring quality, safety, and efficacy.
Our leadership team spearheads scientific advancements and manufacturing excellence, driving Shilpa Biologicals towards a future of impactful healthcare solutions.
Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
+91-836 248 5555
madhav@vbshilpa.com
If you are seeking reliable Ranibizumab manufacturers in Arunachal Pradesh, Shilpa Biologicals is your trusted partner. With a cutting-edge R&D and manufacturing facility in Dharwad, Karnataka, Shilpa Biologicals excels in developing advanced biologics and biosimilars that address critical healthcare needs, including treatments for retinal diseases and other ophthalmic conditions.
At Shilpa Biologicals, we focus on immunology and haematology with a vision to make breakthrough therapies affordable and accessible worldwide. Our scientifically driven approach leverages expertise in molecular biology, biochemistry, immunology, and formulations to develop a robust pipeline of biosimilars and biologics.
Ranibizumab, a vital treatment for age-related macular degeneration and diabetic retinopathy, is part of the innovative biosimilars portfolio we are proud to manufacture, ensuring quality, safety, and efficacy for patients in Arunachal Pradesh and beyond.
Apart from Ranibizumab, we manufacture ORIADALI (Adalimumab 40 mg/0.4 mL), a pre-filled syringe biosimilar designed for subcutaneous use, highlighting our expertise in delivering precise and reliable biologic treatments.
Together, our leaders ensure Shilpa Biologicals remains at the forefront of biosimilar manufacturing innovation.
Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
+91-836 248 5555
madhav@vbshilpa.com
Shilpa Biologicals, headquartered in Dharwad, Karnataka, is a premier biologics manufacturing and services company specializing in the development of curative therapies. With a cutting-edge R&D facility and a team of committed scientists and professionals, we are at the forefront of adenoviral vaccine development and biologics innovation in India.
Our focus on immunology and haematology sectors enables us to develop advanced biologics and vaccine candidates, including adenoviral vector vaccines designed to combat infectious diseases effectively. Leveraging deep scientific knowledge in molecular biology, biochemistry, immunology, and pharmaceutical formulations, we ensure high-quality, scalable vaccine production tailored for global markets.
Shilpa Biologicals’ product pipeline includes biosimilars and biologics such as ORIADALI (Adalimumab 40 mg/0.4 mL), reflecting our commitment to quality and precision in biologic therapies. Our expertise extends seamlessly into adenoviral vaccine manufacturing, ensuring reliability and excellence.
Our leaders bring vision and scientific rigor, guiding Shilpa Biologicals to excel in vaccine development and biologics manufacturing.
Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
+91-836 248 5555
madhav@vbshilpa.com
Shilpa Biologicals, headquartered in Dharwad, Karnataka, is a leading biologics manufacturing and services company specializing in the development of innovative therapies, including albumin and other blood-product segments. With a state-of-the-art R&D and manufacturing facility, we focus on developing affordable and effective treatments for immunology and haematology sectors.
Our scientific strength lies in leveraging advanced molecular biology, biochemistry, immunology, and formulation technologies to develop high-quality albumin products. Shilpa Biologicals has built a robust pipeline of wholly owned projects aimed at revolutionizing the blood-product segment with precision-engineered albumin formulations, catering to both domestic and global markets.
In addition to albumin, Shilpa Biologicals manufactures ORIADALI (Adalimumab 40 mg/0.4 mL), a pre-filled syringe biosimilar reflecting our expertise in biologic therapies and commitment to treatment reliability and patient safety.
Our leadership drives innovation and operational excellence to maintain Shilpa Biologicals’ status as a top-tier albumin development manufacturer in India.
Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
+91-836 248 5555
madhav@vbshilpa.com
Shilpa Biologicals, with its cutting-edge R&D and manufacturing facility located in Dharwad, Karnataka, is a pioneering biologics manufacturing and services company dedicated to developing curative therapies. We specialize in conducting comprehensive scaleup studies that help transition laboratory processes to commercial production seamlessly.
Our expertise lies in leveraging a strong foundation of molecular biology, biochemistry, immunology, and pharmaceutical formulations to perform detailed scaleup studies. These studies ensure process optimization, quality consistency, and regulatory compliance — critical for successful biologics manufacturing.
At Shilpa Biologicals, innovation drives our ability to disrupt traditional pharmaceutical development. Our talented professionals and scientists continuously strive to provide next-generation solutions to complex scientific challenges, ensuring that scaleup processes meet the highest standards.
We proudly manufacture ORIADALI (Adalimumab 40 mg/0.4 mL), a precision-engineered pre-filled syringe biosimilar, exemplifying our manufacturing quality and reliability.
Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
+91-836 248 5555
madhav@vbshilpa.com
Shilpa Biologicals, a trusted name in the biologics manufacturing industry, is dedicated to developing advanced and affordable curative therapies that make a real difference in patients' lives worldwide. Though our state-of-the-art R&D and manufacturing facility is located in Dharwad, Karnataka, we proudly extend our Contract Development and Manufacturing Organization (CDMO) biologics services to clients across India, including Arunachal Pradesh.
As one of India’s premier CDMO biologics manufacturers, Shilpa Biologicals specializes in providing comprehensive development and manufacturing solutions for biologic therapies. Our core strengths lie in:
With a strong focus on immunology and haematology, we have built a robust pipeline of innovative therapies aimed at making treatments accessible and affordable.
✅ World-Class R&D Facility: Advanced infrastructure with cutting-edge technology located in Dharwad, Karnataka.
✅ Expert Team: Highly skilled scientists, researchers, and manufacturing professionals dedicated to scientific excellence.
✅ Innovation Driven: Our talented team is committed to solving complex scientific challenges with novel solutions.
✅ Global Competence: Our biologics are designed to meet international standards, competing globally for best-in-class quality.
Experience precision biologic care with ORIADALI (Adalimumab 40 mg/0.4 mL), a pre-filled syringe intended for subcutaneous use. Manufactured with uncompromised quality by Shilpa Biosciences, ORIADALI reflects our commitment to patient care and therapeutic innovation.
At Shilpa Biologicals, we offer end-to-end CDMO services covering:
Our capabilities empower pharmaceutical and biotech companies in Arunachal Pradesh to bring their biologic products from concept to market faster, with enhanced quality and efficiency.
Our success stems from visionary leadership and a commitment to innovation:
If you are looking for reliable and innovative CDMO biologics manufacturers in Arunachal Pradesh, Shilpa Biologicals is your trusted partner. We combine scientific excellence with manufacturing expertise to help you achieve success in the biologics market.
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
Call us: +91-836-248-5555
Email: madhav@vbshilpa.com
Let us help you bring breakthrough therapies to life.
Shilpa Biologicals, based in Dharwad, Karnataka, is a leading name among protein characterization manufacturers in Karnataka, dedicated to advancing biologics research and development. With a world-class R&D and manufacturing facility, we specialize in delivering high-quality protein characterization services that drive innovation in the biopharmaceutical industry.
At Shilpa Biologicals, we understand that protein characterization is the cornerstone of developing safe, effective, and high-quality biologic therapies. Our expert team of scientists, with deep knowledge in molecular biology, biochemistry, immunology, and formulations, ensures that every protein is analyzed, validated, and optimized with utmost precision.
Our services support the development of complex biologics, including biosimilars and innovative therapeutic proteins, with a focus on immunology and haematology.
✅ Primary and Secondary Structure Analysis
✅ Glycosylation Profiling
✅ Aggregation and Stability Studies
✅ Potency and Bioactivity Assays
✅ Immunogenicity Assessments
✅ Formulation Development
Our rigorous analytical approaches meet international quality standards, supporting pharmaceutical companies in Karnataka and beyond to bring safe and effective biologic products to market.
Our dedicated R&D team continues to push the boundaries of protein science. Through advanced protein characterization, we support the development of novel biologics such as ORIADALI (Adalimumab 40 mg/0.4 mL)—a pre-filled syringe for subcutaneous use, manufactured with precision by Shilpa Biosciences.
We believe that science has the power to transform lives, and our mission is to make curative therapies affordable and accessible worldwide.
Guided by visionary leadership, Shilpa Biologicals continues to excel in biologics innovation:
If you are seeking reliable and innovative protein characterization manufacturers in Karnataka, partner with Shilpa Biologicals for end-to-end solutions in biologics development.
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
Phone: +91-836-248-5555
Email: madhav@vbshilpa.com
Shilpa Biologicals – Driving Science, Transforming Lives.
Looking for trusted biologics manufacturers in Assam? Shilpa Biologicals, a leading name in the field of biologics development and manufacturing, brings cutting-edge healthcare solutions to meet the growing demands of the pharmaceutical and biotechnology sectors in Assam and across India.
With a state-of-the-art R&D and manufacturing facility in Dharwad, Karnataka, Shilpa Biologicals focuses on creating curative therapies in the fields of immunology and haematology, with a strong mission to make advanced biologic treatments accessible and affordable worldwide.
Our expertise in biologics manufacturing stems from a deep scientific foundation in:
Through these core competencies, we have built a robust pipeline of innovative biologics and biosimilars designed to meet international quality standards while maintaining cost-effectiveness.
At the forefront of our product innovation is ORIADALI (Adalimumab 40 mg/0.4 mL)—an advanced biologic solution available in a pre-filled syringe for subcutaneous use. Manufactured by Shilpa Biosciences, ORIADALI reflects our commitment to precision care, reliability, and high-quality treatment.
✅ Biosimilar Development and Manufacturing
✅ Protein Characterization & Analytical Services
✅ Regulatory Support
✅ Clinical and Commercial Scale Production
✅ Formulation Development and Fill-Finish Solutions
Our CDMO (Contract Development and Manufacturing Organization) services are designed to help pharmaceutical and biotech companies in Assam accelerate their drug development timelines while ensuring the highest standards of safety and efficacy.
Our leadership team brings vision, expertise, and a passion for innovation:
For reliable and innovative biologics manufacturing services in Assam, connect with Shilpa Biologicals—your trusted partner in driving scientific excellence and improving patient outcomes.
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
Phone: +91-836-248-5555
Email: madhav@vbshilpa.com
Shilpa Biologicals – Advancing Biologics. Transforming Lives.
If you are searching for trusted Ranibizumab manufacturers in Assam, Shilpa Biologicals is a name you can rely on. Headquartered with a cutting-edge R&D and manufacturing facility in Dharwad, Karnataka, we are a leading biologics manufacturing and services company, dedicated to advancing healthcare through innovative, affordable biologic therapies.
Our focus lies in the development and manufacturing of high-quality biosimilars and biologics, including Ranibizumab, to address unmet medical needs and improve patient outcomes both in India and globally.
Ranibizumab is a monoclonal antibody fragment widely used in the treatment of age-related macular degeneration (AMD), diabetic macular edema (DME), and other vision-threatening retinal conditions. At Shilpa Biologicals, we apply deep scientific expertise and cutting-edge manufacturing processes to produce reliable, effective, and affordable Ranibizumab biosimilars.
Our capabilities in protein characterization, formulation development, and regulatory compliance ensure that our products meet global standards of safety, quality, and efficacy.
In addition to Ranibizumab manufacturing, Shilpa Biosciences has introduced ORIADALI (Adalimumab 40 mg/0.4 mL)—an advanced solution for autoimmune disorders, manufactured to the highest quality standards, reflecting our dedication to therapeutic excellence.
✅ Biosimilar Development (including Ranibizumab)
✅ CDMO Services (Contract Development & Manufacturing)
✅ Protein Characterization & Analytical Testing
✅ Formulation & Fill-Finish Solutions
✅ Regulatory Support for Global Markets
We are committed to helping pharmaceutical and biotechnology companies in Assam and beyond to bring affordable, life-saving biologics to patients faster and more efficiently.
Our success is driven by visionary leaders and a dedicated team of experts:
At Shilpa Biologicals, we are dedicated to advancing Ranibizumab manufacturing and other biologics to improve patient care across India, including in Assam. Our scientific rigor, manufacturing excellence, and patient-centric approach make us a preferred partner for biosimilar development.
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
Call us: +91-836-248-5555
Email: madhav@vbshilpa.com
Shilpa Biologicals – Pioneering Biologics. Transforming Healthcare.
Looking for reliable CMO biologics manufacturers in Bihar? Shilpa Biologicals, headquartered with a cutting-edge R&D and manufacturing facility in Dharwad, Karnataka, is a leading biologics manufacturing and services company dedicated to advancing healthcare with innovative, high-quality biologic solutions.
As a trusted Contract Manufacturing Organization (CMO), we offer end-to-end biologics development and manufacturing services, helping pharmaceutical and biotech companies in Bihar and across India bring life-changing therapies to market efficiently and cost-effectively.
At Shilpa Biologicals, our services are powered by a highly experienced team of scientists, industry experts, and manufacturing professionals who are committed to delivering excellence in every project. Our expertise spans across:
✅ Biosimilar Development & Manufacturing
✅ Process Development & Scale-Up
✅ Protein Characterization & Analytical Services
✅ Formulation Development & Fill-Finish Services
✅ Regulatory Compliance & Support
Our biologics pipeline focuses primarily on immunology and haematology, with the mission to make advanced therapies affordable and accessible to patients worldwide, including those in Bihar.
In addition to our CMO services, Shilpa Biosciences has introduced ORIADALI (Adalimumab 40 mg/0.4 mL)—an advanced solution for autoimmune conditions. This pre-filled syringe reflects our commitment to precision care, quality, and patient safety.
Our journey is guided by visionary leadership and scientific integrity:
If you are seeking an experienced and innovative CMO biologics manufacturer in Bihar, Shilpa Biologicals offers the expertise, infrastructure, and commitment you need to succeed in the fast-evolving biologics landscape.
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
Phone: +91-836-248-5555
Email: madhav@vbshilpa.com
Shilpa Biologicals – Empowering Biologics. Advancing Healthcare.
When it comes to advanced biologics manufacturing and innovation, Shilpa Biologicals stands out as one of the leading biologicals companies in India, proudly extending its world-class services to clients in Andhra Pradesh and across the nation. With a cutting-edge R&D and manufacturing facility located in Dharwad, Karnataka, Shilpa Biologicals is dedicated to developing affordable and effective curative therapies that transform patient care globally.
At Shilpa Biologicals, our mission is to make life-saving biologic therapies accessible and affordable for patients everywhere. We specialize in the research, development, and manufacturing of high-quality biosimilars and biologic products with a strong focus on immunology and haematology.
Our expertise is rooted in deep scientific knowledge across:
We have built a robust pipeline of innovative biologics designed to meet global standards, and our services are trusted by pharmaceutical companies, healthcare providers, and research institutions, including those in Andhra Pradesh.
✅ Biosimilar Development and Manufacturing
✅ CDMO (Contract Development and Manufacturing Organization) Services
✅ Protein Characterization & Analytical Solutions
✅ Formulation Development & Fill-Finish Services
✅ Regulatory Compliance & Support
Our team of dedicated scientists and industry professionals is committed to delivering innovative solutions that meet the highest standards of safety, quality, and efficacy.
Our portfolio features ORIADALI (Adalimumab 40 mg/0.4 mL)—a pre-filled syringe designed for subcutaneous use, reflecting our dedication to precision care and advanced treatment options in autoimmune diseases.
Manufactured with stringent quality controls, ORIADALI represents our passion for bringing reliable, high-quality biologic therapies to the market.
Our journey is guided by a team of visionary leaders who continue to propel Shilpa Biologicals to the forefront of biologics innovation:
Whether you are looking for biosimilar development, CDMO services, or innovative biologic solutions, Shilpa Biologicals is your trusted partner in Andhra Pradesh. We combine scientific excellence with manufacturing expertise to help bring transformative therapies to market.
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
Call us: +91-836-248-5555
Email: madhav@vbshilpa.com
Shilpa Biologicals – Pioneering Science, Advancing Healthcare.
Shilpa Biologicals, with its state-of-the-art R&D and manufacturing facility in Dharwad, Karnataka, is one of the trusted perfusion manufacturers in India, dedicated to advancing healthcare through innovative biologics and cutting-edge technologies. Our mission is to develop curative therapies that are not only effective but also affordable for patients across India and around the globe.
With a strong foundation in molecular biology, biochemistry, immunology, and pharmaceutical formulations, we specialize in creating high-quality biologics and biosimilars that contribute to the progress of modern medicine, including solutions related to perfusion-based applications.
Perfusion plays a crucial role in various medical treatments, especially in organ preservation, tissue engineering, and advanced drug delivery systems. At Shilpa Biologicals, our dedicated team of scientists and industry professionals brings together extensive expertise and innovation to meet the complex demands of this sector.
Our biologics and manufacturing capabilities align with the requirements of perfusion technology, ensuring reliability, safety, and global quality standards.
✅ Advanced Scientific Expertise: Powered by deep knowledge in molecular biology, immunology, and biochemistry.
✅ World-Class Manufacturing: Cutting-edge infrastructure in Dharwad, Karnataka, enabling precision manufacturing and high scalability.
✅ Quality & Compliance: Stringent adherence to global regulatory standards to ensure product safety and efficacy.
✅ Innovative Product Pipeline: Focused on immunology, haematology, and novel biologics that transform patient care.
We are proud to introduce ORIADALI (Adalimumab 40 mg/0.4 mL)—a pre-filled syringe designed for subcutaneous use, manufactured with the highest quality standards by Shilpa Biosciences. ORIADALI reflects our unwavering commitment to delivering precision biologics that improve patients' quality of life.
Our integrated services support the development of next-generation perfusion solutions and biologic therapies that meet the diverse needs of the healthcare industry.
Our growth and innovation are driven by a visionary leadership team dedicated to scientific excellence and healthcare transformation:
If you are looking for reliable perfusion manufacturers in India, Shilpa Biologicals is your trusted partner. We combine innovation, scientific expertise, and manufacturing excellence to help you bring high-quality healthcare solutions to market.
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
Call us: +91-836-248-5555
Email: madhav@vbshilpa.com
Shilpa Biologicals – Powering Innovation. Advancing Healthcare.
Shilpa Biologicals stands at the forefront of biologics manufacturing in India, dedicated to developing innovative, curative therapies that aim to transform healthcare for patients around the world. With a state-of-the-art R&D and manufacturing facility located in Dharwad, Karnataka, Shilpa Biologicals combines scientific excellence with a passion for making life-saving treatments accessible and affordable.
Our cutting-edge team of scientists, researchers, and manufacturing professionals work relentlessly to deliver best-in-class biologics solutions. At Shilpa Biologicals, we specialize in the development and production of therapies in the immunology and haematology sectors. We are driven by a single goal — to improve patient outcomes through innovation and affordability.
By leveraging advanced expertise in molecular biology, biochemistry, immunology, and pharmaceutical formulations, we have built a strong portfolio of wholly owned biologics projects that have the potential to revolutionize the blood-product market. Our commitment to research and development allows us to compete globally while maintaining the highest standards of quality and compliance.
One of our flagship products, ORIADALI (Adalimumab 40 mg/0.4 mL), is a precision-designed pre-filled syringe for subcutaneous use, offering reliable and high-quality treatment for patients with autoimmune conditions. Manufactured with strict quality controls at Shilpa Biosciences, ORIADALI embodies our promise of excellence, safety, and efficacy in biologics manufacturing.
As a leading CDMO (Contract Development and Manufacturing Organization) in India, Shilpa Biologicals offers end-to-end services that cater to diverse biopharmaceutical needs. Our CDMO capabilities include:
We help bring biologics products to market faster and more efficiently, thanks to our highly specialized infrastructure and expert team.
The visionary leadership team at Shilpa Biologicals ensures we remain at the cutting edge of innovation:
Our leaders drive Shilpa Biologicals with a clear mission: to harness science for the betterment of global healthcare.
We are proud to be recognized as one of the top biologics manufacturers in India, committed to delivering solutions that improve lives and redefine healthcare possibilities.
Location:
Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, INDIA
Call Us: +91-836-248-5555
Email: madhav@vbshilpa.com
Shilpa Biologicals — Advancing Healthcare, Transforming Lives.
Shilpa Biologicals, based in Dharwad, Karnataka, is one of the leading CDMO biologics manufacturers in India, dedicated to advancing healthcare through innovation and affordable biologic solutions. With a state-of-the-art R&D and manufacturing facility, we specialize in developing curative therapies in the fields of immunology and haematology, aiming to make advanced treatments accessible to patients worldwide.
At Shilpa Biologicals, our strength lies in a passionate team of scientists and professionals committed to excellence in biologics manufacturing and services. By leveraging expertise in molecular biology, biochemistry, immunology, and formulations, we have built a robust pipeline of projects that hold the potential to revolutionize the blood-product segment and position India on the global stage of biopharmaceutical innovation.
Our approach combines scientific innovation with patient-centric care, ensuring that every product we develop meets international standards of quality, safety, and efficacy.
As a trusted biologics CDMO in India, we provide end-to-end Contract Development and Manufacturing Organization (CDMO) services, including:
With our strong scientific foundation and cutting-edge infrastructure, we support pharmaceutical companies and biotech innovators in accelerating their product development while ensuring cost-effectiveness and reliability.
Our pipeline is focused on biosimilars in immunology and haematology, designed to address critical patient needs. One of our key innovations includes:
ORIADALI – Advanced Adalimumab Solution for Injection
ORIADALI represents our commitment to delivering high-quality, affordable biosimilars that improve patient outcomes.
Shilpa Biologicals is guided by visionary leaders driving innovation and excellence:
Their collective expertise ensures that Shilpa Biologicals remains at the forefront of biologics research, biosimilar development, and CDMO partnerships.
Plot 532/A, Belur Industrial Area, Dharwad – 580011, Karnataka, INDIA
Call: +91-836 248 5555
Email: madhav@vbshilpa.com
Shilpa Biologicals, headquartered in Dharwad, Karnataka, is one of the leading biologicals companies in India, dedicated to developing innovative and affordable therapies in immunology and haematology. With a world-class R&D and manufacturing facility, we focus on building transformative solutions that address some of the most complex medical challenges faced by patients across the globe.
At Shilpa Biologicals, we combine scientific excellence with a mission-driven approach. Our strength lies in a team of highly skilled scientists and professionals who continuously push boundaries in biologics manufacturing. Leveraging deep expertise in molecular biology, biochemistry, immunology, and formulations, we have established a robust product pipeline capable of revolutionizing the blood-product segment.
Our commitment is to ensure that life-saving therapies remain accessible, reliable, and affordable for patients worldwide.
Introducing ORIADALI (Adalimumab 40 mg/0.4 mL) – an advanced biologic solution developed with precision.
ORIADALI reflects our dedication to creating world-class biosimilars that redefine treatment possibilities in immunology.
As a trusted partner in the global biopharma space, we offer:
Guided by visionary leaders, Shilpa Biologicals continues to grow as a pioneer in India’s biologics sector:
Their expertise and strategic vision ensure that ShilpaBio remains a leader in biologics innovation and global partnerships.
Plot 532/A, Belur Industrial Area, Dharwad – 580011, Karnataka, INDIA
Call: +91-836 248 5555
Email: madhav@vbshilpa.com
Shilpa Biologicals, with its R&D and manufacturing facility in Dharwad, Karnataka, is a leading albumin manufacturer in India. Specializing in biologics and blood-based therapies, we focus on producing high-quality, reliable albumin solutions for the immunology and haematology sectors, making advanced therapies affordable and accessible to patients across India and globally, including Andhra Pradesh.
Shilpa Biologicals is a biologics manufacturing and services company driven by scientific innovation and patient-centric care. Our team of dedicated scientists and professionals leverage expertise in molecular biology, biochemistry, immunology, and formulations to develop a robust pipeline of products, including albumin, with the potential to revolutionize the blood-product segment.
We combine cutting-edge R&D with high-standard manufacturing practices to deliver products that meet global quality benchmarks. Our commitment is to ensure reliable and safe therapeutics for hospitals, clinics, and patients.
Shilpa Biologicals offers high-quality albumin that is manufactured under strict quality controls to ensure:
Our albumin products support treatments in critical care, immunology, and haematology, offering dependable solutions for healthcare providers throughout Andhra Pradesh.
We also specialize in biosimilars, including ORIADALI (Adalimumab 40 mg/0.4 mL), a pre-filled syringe for subcutaneous use, reflecting our commitment to precision care and advanced biologic therapies.
Our experienced leadership drives Shilpa Biologicals toward innovation and global recognition:
Their guidance ensures excellence in manufacturing, product development, and global collaborations.
Plot 532/A, Belur Industrial Area, Dharwad – 580011, Karnataka, INDIA
Call: +91-836 248 5555
Email: madhav@vbshilpa.com
Shilpa Biologicals, headquartered with its R&D and manufacturing facility in Dharwad, Karnataka, is a premier biologics manufacturing and services company in India. With a strong focus on developing curative therapies, Shilpa Biologicals combines cutting-edge scientific research with a mission to make advanced treatments affordable for patients worldwide.
Shilpa Biologicals operates at the forefront of immunology and haematology research. Our expert team of scientists and committed professionals at the manufacturing floor ensures high-quality production while supporting innovative solutions to critical scientific challenges. By leveraging expertise in molecular biology, biochemistry, immunology, and formulations, Shilpa Biologicals has established a robust product pipeline that is poised to revolutionize the blood-product segment and compete globally.
Our services are driven by a deep confidence in scientific strength, with the goal of making a tangible impact on the global healthcare landscape. At Shilpa Biologicals, talented professionals continuously strive to innovate, ensuring high-quality biologics and advanced therapies reach patients safely and effectively.
Shilpa Biosciences introduces ORIADALI, an advanced Adalimumab (40 mg/0.4 mL) solution in a pre-filled syringe for subcutaneous use. ORIADALI combines precision, reliability, and quality, ensuring effective treatment outcomes for patients.
Shilpa Biologicals is led by an experienced executive and scientific board, driving innovation and growth:
Our leadership team is committed to realizing the full potential of Shilpa Biologicals, ensuring every initiative aligns with patient welfare and global standards.
Shilpa Biologicals offers a wide range of innovative solutions:
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India
Phone: +91-836 248 5555
Email: madhav@vbshilpa.com
Shilpa Biologicals is committed to scientific excellence, innovation, and patient-centric therapies, making it a trusted leader in the biologics industry in India.
Shilpa Biologicals, with its R&D and manufacturing facility in Dharwad, Karnataka, is a leading biologics manufacturing and services company in India. With a focus on developing curative therapies, Shilpa Biologicals combines advanced scientific expertise with a commitment to making treatments affordable and accessible worldwide.
Shilpa Biologicals specializes in the immunology and haematology sectors, leveraging a strong foundation in molecular biology, biochemistry, immunology, and formulations. Our talented team of scientists and manufacturing professionals ensures the highest quality of biologics, while our robust product pipeline includes innovative projects capable of revolutionizing the blood-product segment globally.
Our services are rooted in scientific excellence and innovation, allowing us to contribute to breakthrough therapies that address critical healthcare challenges. The company’s professionals are dedicated to developing next-generation solutions for deep scientific problems, ensuring global competitiveness and patient-centric outcomes.
Shilpa Biosciences also introduces ORIADALI (Adalimumab 40 mg/0.4 mL), a pre-filled syringe for subcutaneous use. This solution demonstrates our commitment to precision, reliability, and quality in biologic therapies.
Shilpa Biologicals’ growth and innovation are driven by a highly experienced executive board and scientific leadership team:
Our leadership is committed to unlocking the full potential of Shilpa Biologicals, ensuring high-quality biologics and services reach patients worldwide.
Shilpa Biologicals offers a wide range of advanced biologics and contract services:
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India
Phone: +91-836 248 5555
Email: madhav@vbshilpa.com
Shilpa Biologicals stands as a trusted leader among Ranibizumab manufacturers in India, delivering scientifically advanced, reliable, and affordable biologic therapies to patients worldwide.
Shilpa Biologicals, with its R&D and manufacturing facility in Dharwad, Karnataka, is a leading biologics manufacturing and services company in India. We specialize in developing curative therapies with a mission to make advanced treatments affordable and accessible to patients worldwide.
Shilpa Biologicals focuses on the immunology and haematology sectors, leveraging expertise in molecular biology, biochemistry, immunology, and formulations. Our talented scientists and manufacturing professionals work tirelessly to ensure high-quality biologics, while our robust product pipeline includes innovative projects capable of revolutionizing the blood-product segment globally.
With a foundation in scientific excellence and innovation, Shilpa Biologicals contributes to breakthrough therapies that address critical healthcare challenges. Our professionals are committed to thinking beyond the status quo and developing next-generation solutions for complex scientific problems.
Shilpa Biosciences also introduces ORIADALI (Adalimumab 40 mg/0.4 mL), a pre-filled syringe designed for subcutaneous use. ORIADALI ensures precision, reliability, and quality in treatment, reflecting Shilpa’s commitment to patient-centered therapies.
Shilpa Biologicals is led by an experienced executive board and scientific leadership team:
Our leaders ensure Shilpa Biologicals continues to deliver high-quality, globally competitive biologics and solutions for patients worldwide.
Shilpa Biologicals provides a broad range of advanced biologics and CDMO services:
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India
Phone: +91-836 248 5555
Email: madhav@vbshilpa.com
Shilpa Biologicals is a trusted name among Tenecteplase manufacturers in Gujarat, offering scientifically advanced, reliable, and affordable biologic therapies to patients worldwide.
Shilpa Biologicals, with its R&D and manufacturing facility in Dharwad, Karnataka, is a leading biosimilars manufacturer in India, providing high-quality biologics and services with a focus on curative therapies. Our mission is to make advanced treatments affordable and accessible to patients worldwide.
Shilpa Biologicals specializes in the immunology and haematology sectors, leveraging expertise in molecular biology, biochemistry, immunology, and formulations. Our talented team of scientists and manufacturing professionals ensures high-quality biosimilars, while our robust product pipeline includes innovative projects capable of revolutionizing the blood-product segment and competing globally.
With a strong foundation in scientific excellence and innovation, Shilpa Biologicals is committed to developing next-generation solutions that address complex healthcare challenges. Our professionals are driven to push the boundaries of research, ensuring cutting-edge therapies reach patients safely and effectively.
Shilpa Biosciences introduces ORIADALI (Adalimumab 40 mg/0.4 mL), a pre-filled syringe for subcutaneous use. ORIADALI is designed for precision, reliability, and quality, reflecting Shilpa’s commitment to patient-centric biologic therapies.
Shilpa Biologicals’ growth and innovation are driven by a highly experienced executive board and leadership team:
Our leaders ensure that Shilpa Biologicals continues to deliver globally competitive biosimilars and advance scientific innovation.
Shilpa Biologicals offers a wide range of biosimilars and CDMO services:
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India
Phone: +91-836 248 5555
Email: madhav@vbshilpa.com
Shilpa Biologicals is a trusted name among biosimilar manufacturers in Goa, delivering innovative, reliable, and affordable biologic therapies to patients worldwide.
Shilpa Biologicals, with its R&D and manufacturing facility in Dharwad, Karnataka, is a leading biologics manufacturing and services company in India. With a strong focus on developing curative therapies, Shilpa Biologicals aims to make advanced biologic treatments affordable and accessible to patients worldwide.
Shilpa Biologicals specializes in the immunology and haematology sectors, combining expertise in molecular biology, biochemistry, immunology, and formulations. Our dedicated team of scientists and professionals ensures high-quality biologics, while our robust product pipeline includes projects capable of revolutionizing the blood-product segment and competing globally.
Rooted in scientific innovation and excellence, Shilpa Biologicals delivers therapies that address critical healthcare challenges. Our professionals are committed to thinking beyond conventional solutions, creating next-generation biologics with potential global impact.
Shilpa Biosciences introduces ORIADALI (Adalimumab 40 mg/0.4 mL), a pre-filled syringe for subcutaneous use. ORIADALI exemplifies Shilpa’s commitment to precision, reliability, and quality in biologic therapies.
Shilpa Biologicals’ leadership drives innovation and global competitiveness:
Their guidance ensures Shilpa Biologicals consistently delivers world-class biologics and services.
Shilpa Biologicals offers a broad range of biologics and CDMO services:
Address: Plot 532/A, Belur Industrial Area, Dharwad-580011, Karnataka, India
Phone: +91-836 248 5555
Email: madhav@vbshilpa.com
Shilpa Biologicals is a trusted name among Aflibercept manufacturers in Gujarat, providing scientifically advanced, reliable, and affordable biologic therapies to patients worldwide.

